Answered step by step
Verified Expert Solution
Question
1 Approved Answer
8 A compound made of two elements, Iridium( Ir, MM =192.2 g/mol) and oxygen (O, MM=16.00 g/mol), was produced in a lab by heating iridium
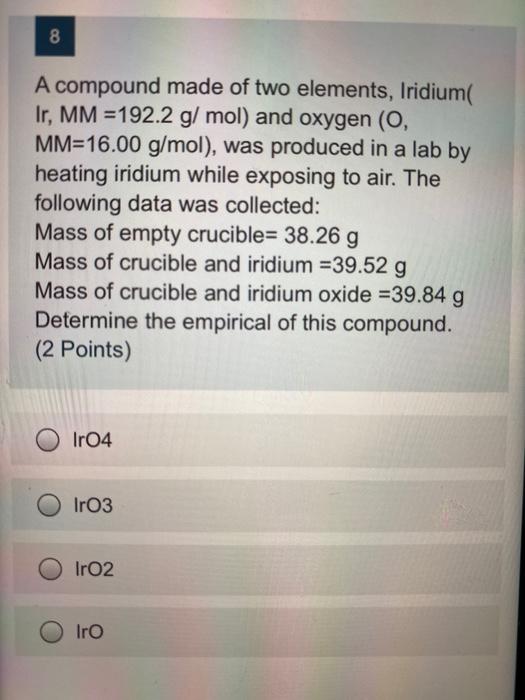 8 A compound made of two elements, Iridium( Ir, MM =192.2 g/mol) and oxygen (O, MM=16.00 g/mol), was produced in a lab by heating iridium while exposing to air. The following data was collected: Mass of empty crucible= 38.26 g Mass of crucible and iridium =39.52 g Mass of crucible and iridium oxide =39.84 g Determine the empirical of this compound. (2 Points) Ir04 Iro3 Iro2 Iro
8 A compound made of two elements, Iridium( Ir, MM =192.2 g/mol) and oxygen (O, MM=16.00 g/mol), was produced in a lab by heating iridium while exposing to air. The following data was collected: Mass of empty crucible= 38.26 g Mass of crucible and iridium =39.52 g Mass of crucible and iridium oxide =39.84 g Determine the empirical of this compound. (2 Points) Ir04 Iro3 Iro2 Iro

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


