Answered step by step
Verified Expert Solution
Question
1 Approved Answer
8.6 g of helium gas follows the process 1-2-3 shown in the figure (Figure 1). Figure P of 2 atm P2 657C- 37C- 2
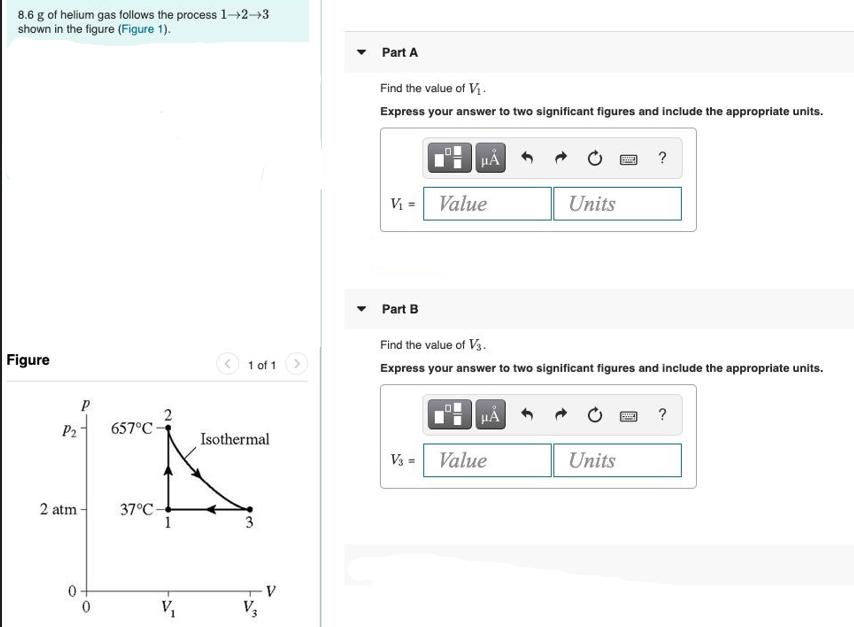
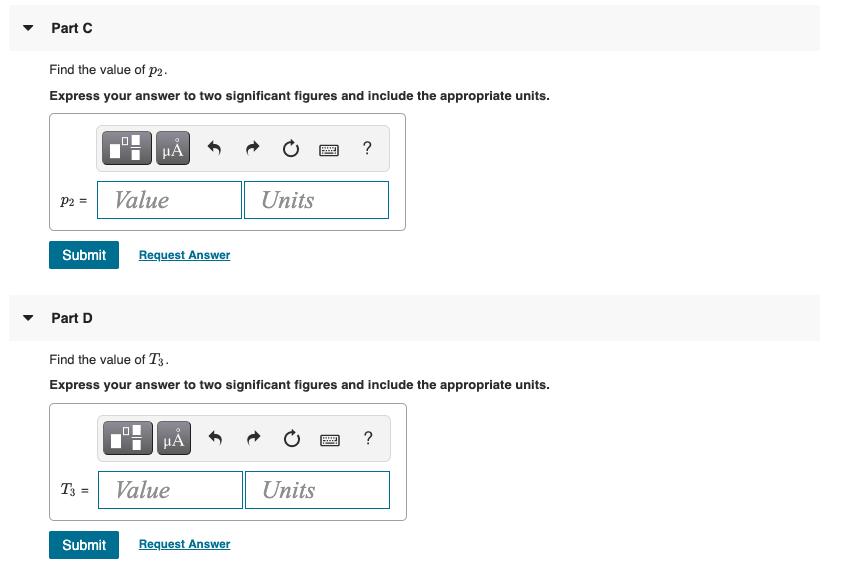
8.6 g of helium gas follows the process 1-2-3 shown in the figure (Figure 1). Figure P of 2 atm P2 657C- 37C- 2 V 1 of 1 Isothermal 3 Part A Find the value of V. Express your answer to two significant figures and include the appropriate units. V = Value Part B H V3 = Find the value of V3. Express your answer to two significant figures and include the appropriate units. H Units Value ? Units ? Part C Find the value of p2. Express your answer to two significant figures and include the appropriate units. P2 = Part D Submit Request Answer A T3 = Value Submit Find the value of T3. Express your answer to two significant figures and include the appropriate units. A Value Units Request Answer Units ? d ?
Step by Step Solution
There are 3 Steps involved in it
Step: 1
Given figure P P2 657d 29th 0 2 37c 3 T 37 c 310 k T2657C T 657c 930 k T3 T...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


