9. What is the coordination entity formed when excess of aqueous KCN is added to an...
Fantastic news! We've Found the answer you've been seeking!
Question:
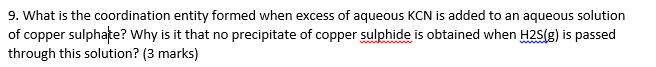
Transcribed Image Text:
9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution? (3 marks) 9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution? (3 marks)
Expert Answer:
Answer rating: 100% (QA)
Answer P generation F generation RY Ry rY ry RY RRYY Round yellow RRYY Round yellow ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the coordination number of each sphere in (a) A simple cubic cell, (b) A body-centered cubic cell, (c) A face-centered cubic cell? Assume the spheres are all the same?
-
Ammonia and potassium iodide solutions are added to an aqueous solution of Cr(NO 3 ) 3 . A solid is isolated (compound A), and the following data are collected: i. When 0.105 g of compound A was...
-
What is the coordination number of cobalt in the complex [Co(en)(NH 3 )2Cl 2 ]ClO 4 ? a. 2 b. 3 c. 4 d. 5 e. 6
-
Determine if the following strains satisfy the compatibility equations (2.6.2): a. b. c. where A, B and C are constants. Equation 2.6.2 ex=Ay, ey = ez = 0, exy = (Ax+Bz)/2, eyz = Bxz + Cy, ezx = C.x
-
A 20-year maturity bond with par value $1,000 makes semiannual coupon payments at a coupon rate of 8%. Find the bond equivalent and effective annual yield to maturity of the bond if the bond price...
-
What is the purpose for determining a set of behavior categories and creating a list of specific behaviors to define each category before making behavioral observation?
-
Under CCP 581(b)(1), barring certain exceptions, without a court order a plaintiff has an absolute right to dismiss his or her complaint any time before the commencement of trial. Further investigate...
-
Management of Shreveport Community Hospital has decided to allocate the budgeted costs of its three support departments (administration, public relations, and maintenance) to its three operating...
-
First and second mortgages may potentially provide relatively high rates of return due to their special risk characteristics
-
The Hatcher Company is in the process of developing a new inventory management system. One of the event handling processes in that system is Receive Supplier Shipments. The (inexperienced) systems...
-
Write a single line of ARM assembly code to set bits 7, 10, and 12 of register r9. Write a single line of ARM assembly code to invert the lower six bits of register r1. 9. a) What are the contents of...
-
Describe one to two differences between single vendor solutions and multiple vendor integrated solutions. What is the role and importance of multiplatform integration in health care IT systems?...
-
Shellfish Co makes four components, W, X, Y and Z, for which costs in the forthcoming year are expected to be as follows. X Y 2 Production (units) 1,000 2.000 4,000 3,000 Unit marginal costs $ $ $ $...
-
The income statement and additional data of Newton Travel Products, Inc., follow: (Click the icon to view the income statement.) Requirements (Click the icon to view the additional data.) 1. Prepare...
-
Arbagugu Products Share Co makes outdoor shirts. Data relating to the coming year's planned operations are as follows. Sales (230,000 shirts) $4,140,000 Cost of goods sold 2,760,000 Gross profit...
-
9. What is the maturity value of a $54,600, 4%, 6-month Note Receivable?
-
21. What is the net present value of the flier project, which is a 3-year project where Dispersion would spread fliers all over Fairfax? The project would involve an initial investment in equipment...
-
Show that if A is any m n matrix, then Im A = A and AIn = A.
-
(a) What is the frequency of light having a wavelength of 456 nm? (b) What is the wavelength (in nanometers) of radiation having a frequency of 2.45 109 Hz? (This is the type of radiation used in...
-
At STP, 0.280 L of a gas weighs 0.400 g. Calculate the molar mass of the gas.
-
Because of its detrimental effect on the environment, the lead compound described in Problem 3.138 has been replaced in recent years by methyl tert -butyl ether (a compound of C, H, and O) to enhance...
-
The steps, in order, in the decision-making process are (a) identify issue, analyse information, gather information, identify decision-making tool, evaluate results; make a decision. (b) gather...
-
Which of the following is not a typical accountants role? (a) A financial controller. (b) A marketing manager. (c) An auditor. (d) All of the above are roles of an accountant.
-
Describe the financial reporting environment. Indicate whether each statement is true or false. (a) Accounting standards are set by the Financial Reporting Council. (b) The ASX Listing Rules are only...

Study smarter with the SolutionInn App


