Answered step by step
Verified Expert Solution
Question
1 Approved Answer
A 10.00-mL blood sample was diluted to 100.00 mL. 10.00 mL of the resulting solution was analyzed for calcium by precipitating all of the calcium
A 10.00-mL blood sample was diluted to 100.00 mL. 10.00 mL of the resulting solution was analyzed for calcium by precipitating all of the calcium as calcium oxalate, CaC2O4. The solid CaC2O4 was then redissolved in H2SO4 and titrated with 0.00886 M KMnO4. What is the concentration of Ca2+ in the blood sample expressed as milligrams Ca2+/mL blood if the endpoint was reached with the addition of 1.14 mL of the KMnO4 solution?
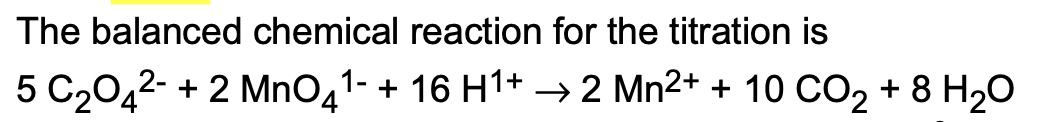
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


