Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The molecule shown below is Efavirenz, which is a reverse transcriptase inhibitor drug used for the treatment of HIV infection. In this question we
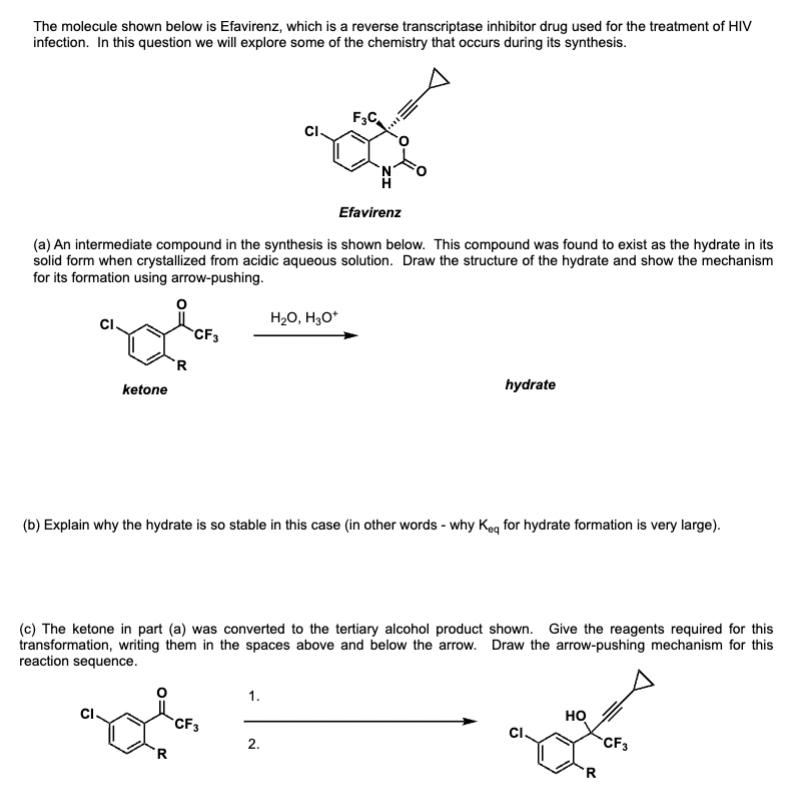
The molecule shown below is Efavirenz, which is a reverse transcriptase inhibitor drug used for the treatment of HIV infection. In this question we will explore some of the chemistry that occurs during its synthesis. F3C Efavirenz (a) An intermediate compound in the synthesis is shown below. This compound was found to exist as the hydrate in its solid form when crystallized from acidic aqueous solution. Draw the structure of the hydrate and show the mechanism for its formation using arrow-pushing. ketone R HO, HO* CF3 hydrate (b) Explain why the hydrate is so stable in this case (in other words - why Keq for hydrate formation is very large). (c) The ketone in part (a) was converted to the tertiary alcohol product shown. Give the reagents required for this transformation, writing them in the spaces above and below the arrow. Draw the arrow-pushing mechanism for this reaction sequence. R CF3 1. 2. HO CF3
Step by Step Solution
There are 3 Steps involved in it
Step: 1
Answer a The structure of the hydrate formed from the intermediate compound is shown below CH3COHCl2 ...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


