Answered step by step
Verified Expert Solution
Question
1 Approved Answer
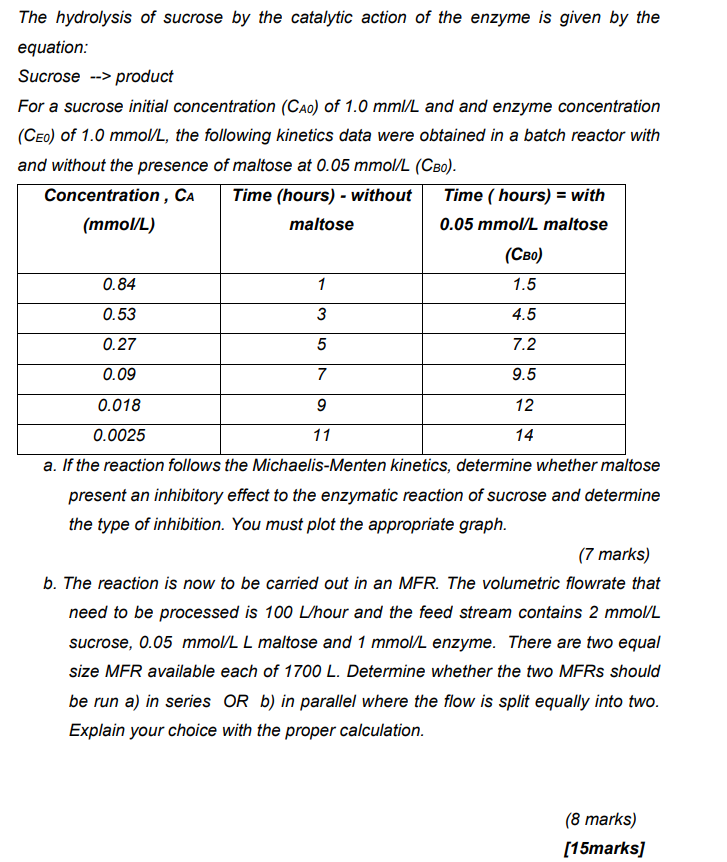
anyone can help me answer asap please The hydrolysis of sucrose by the catalytic action of the enzyme is given by the equation: Sucrose -->
anyone can help me answer asap please
The hydrolysis of sucrose by the catalytic action of the enzyme is given by the equation: Sucrose --> product For a sucrose initial concentration (CAO) of 1.0 mml/L and and enzyme concentration (CEO) of 1.0 mmol/L, the following kinetics data were obtained in a batch reactor with and without the presence of maltose at 0.05 mmol/L (CBo). Concentration, CA Time (hours) - without Time ( hours) = with (mmol/L) maltose 0.05 mmol/L maltose (CBO) 0.84 1 1.5 0.53 3 3 4.5 0.27 5 7.2 0.09 7 9.5 0.018 9 12 0.0025 11 14 a. If the reaction follows the Michaelis-Menten kinetics, determine whether maltose present an inhibitory effect to the enzymatic reaction of sucrose and determine the type of inhibition. You must plot the appropriate graph. (7 marks) b. The reaction is now to be carried out in an MFR. The volumetric flowrate that need to be processed is 100 L/hour and the feed stream contains 2 mmol/L sucrose, 0.05 mmol/L L maltose and 1 mmol/L enzyme. There are two equal size MFR available each of 1700 L. Determine whether the two MFRs should be run a) in series OR b) in parallel where the flow is split equally into two. Explain your choice with the proper calculation. (8 marks) [15marks]Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


