Answered step by step
Verified Expert Solution
Question
1 Approved Answer
1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes

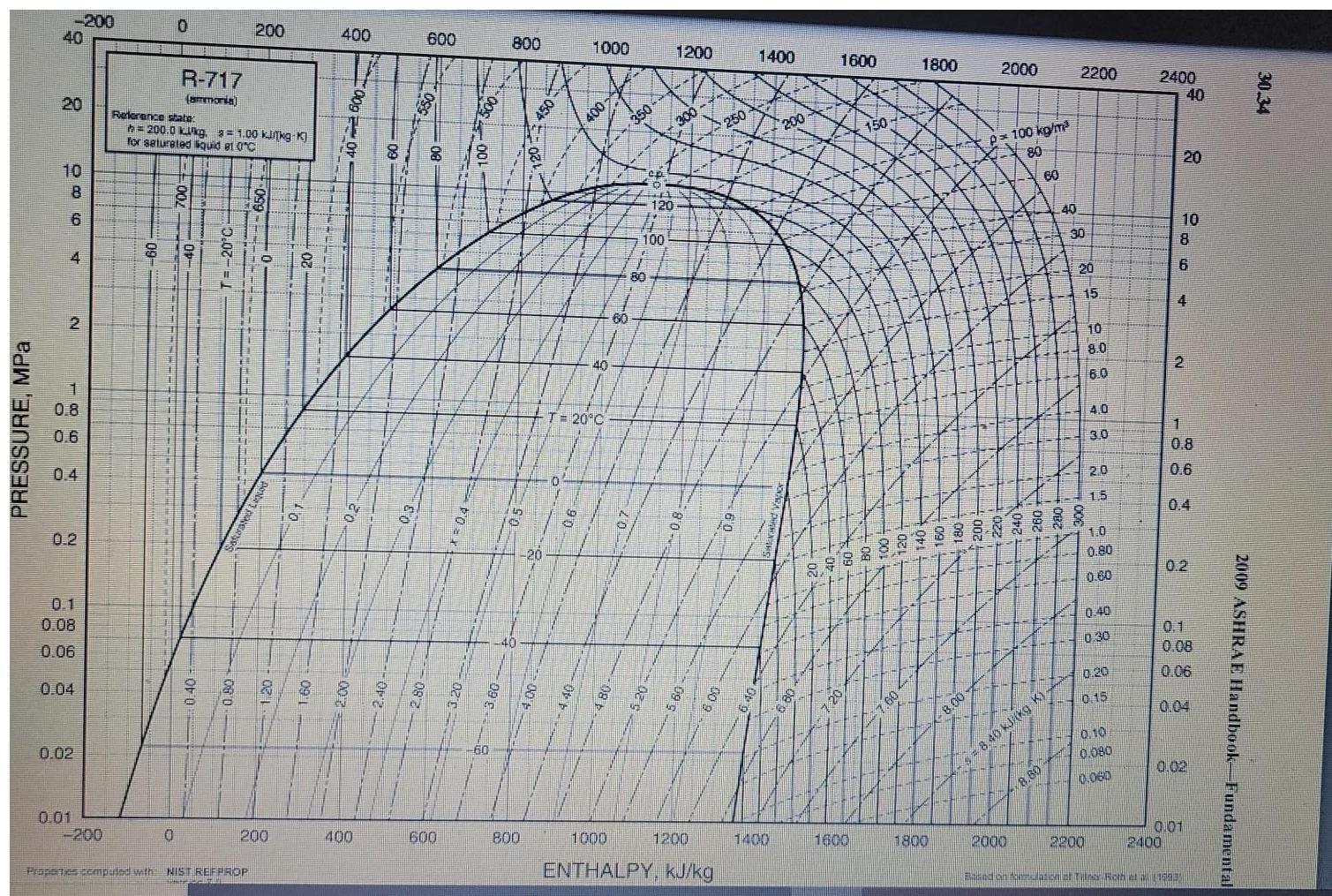
1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes through a throttling valve. Ammonia en ters the compressor at T = 60C and P = 20 bar and is compressed to the pressure P = 200 bar, and then cooled down in the heat exchanger to T3 = 140C and flashed at the pressure P = 1 bar Use the attached PH diagram for ammonia to answer the following questions: A. (10) Draw this process on the attached PH diagram. B. (5) Calculate the fraction of ammonia liquefied. C. (5) Calculate the work performed in the compressor. D. (5) Calculate the amount of heat rejected in the heat exchanger. E. (5) How would the fraction of ammonia liquefied change if the amount of heat rejected in the heat exchanger remained the same, but the compressor is replaced with a non-ideal one? (Briefly explain). PRESSURE, MPa -200 40 20 10 8 6 4 2 1864 0.8 0.6 0.2 0.1 0.08 0.06 0.04 0.01 0.02 -200 60 0 0 R-717 (ammonia) Reference state h=200.0 kkg. = 1.00 kinkg-K] for saturated liquid et 0C 0.40 200 Properties computed with NIST REFPROP O PONA 200 OB 400 20 400 600 8 8 600 500 800 15 6 40 800 450 1000 400 140 T= 20C 250 60 ep 120 EL 100 1200 300 9 1200 3 100 1000 ENTHALPY, kJ/kg 250 1400 1400 200 1600 1600 150 1800 1800 8.00- 2000 p=100 kg/m 80 60 2000 4-5 = 8.40 kJ/(kg K) 40 8.80 2200 30 20 15 10 8.0 6.0 4.0 3.0 2.0 15 1.0 0.80 0.60 2200 0.40 0:30 0.20 0.15 0.10 0.080 0.060 2400 10 8 6 40 20 4 2 2400 1 0.8 06 0.2 0.08 0.06 0.04 0.01 0.02 and on fostulation af Tile Roth (1203 2009 ASHRAE Handbook-Fundamental 30.34
Step by Step Solution
★★★★★
3.54 Rating (158 Votes )
There are 3 Steps involved in it
Step: 1
A The process can be drawn on the PH diagram as follows B The fraction of ammonia liquefied can be c...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


