Question
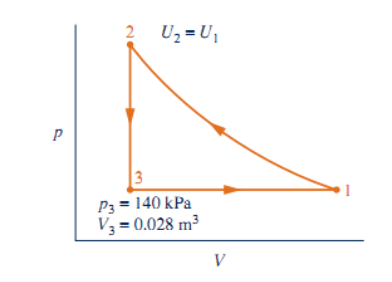
As shown, a gas within a pistoncylinder assembly undergoes a thermodynamic cycle consisting of three processes in series: Process 12: Compression with U2 = U1.
As shown, a gas within a pistoncylinder assembly undergoes a thermodynamic cycle consisting of three processes in series: Process 12: Compression with U2 = U1. Process 23: Constant-volume cooling to p3 = 140 kPa, V3 = 0.028 m3. Process 31: Constant-pressure expansion with W31 = 10.5 kJ. For the cycle, Wcycle = 28.3 kJ. There are no changes in kinetic or potential energy. Determine (a) the volume at state 1, in m3 (b) the work and heat transfer for process 12, each in kJ. (c) Can this be a power cycle? A refrigeration cycle? Explain. (You must identify the correct signs for heat and work)
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


