Answered step by step
Verified Expert Solution
Question
1 Approved Answer
(b) The graph below shows the relationship between enthalpy and temperature of water at 101.325kPa. Determine the enthalpy of saturated liquid water and the change
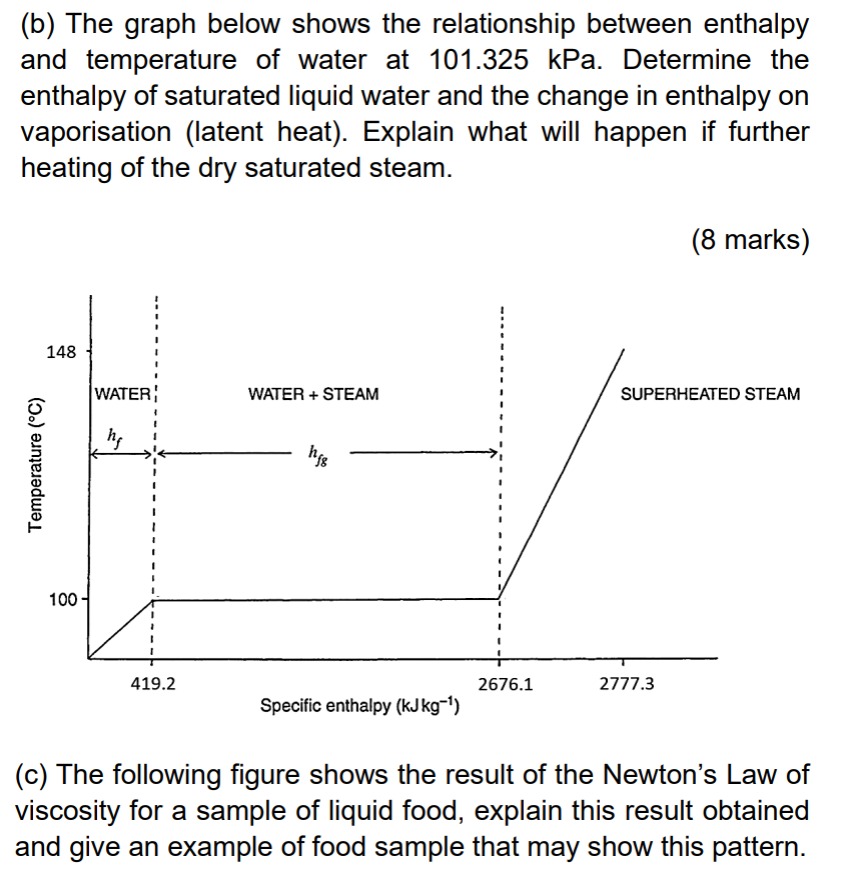 (b) The graph below shows the relationship between enthalpy and temperature of water at 101.325kPa. Determine the enthalpy of saturated liquid water and the change in enthalpy on vaporisation (latent heat). Explain what will happen if further heating of the dry saturated steam. (8 marks) (c) The following figure shows the result of the Newton's Law of viscosity for a sample of liquid food, explain this result obtained and give an example of food sample that may show this pattern
(b) The graph below shows the relationship between enthalpy and temperature of water at 101.325kPa. Determine the enthalpy of saturated liquid water and the change in enthalpy on vaporisation (latent heat). Explain what will happen if further heating of the dry saturated steam. (8 marks) (c) The following figure shows the result of the Newton's Law of viscosity for a sample of liquid food, explain this result obtained and give an example of food sample that may show this pattern Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


