Answered step by step
Verified Expert Solution
Question
1 Approved Answer
c A. 18.30 gram.sample of cobalt is hepted a the preience of exceis suifur. A metal sulfide is formed aith a mass of 33.25 9.
c 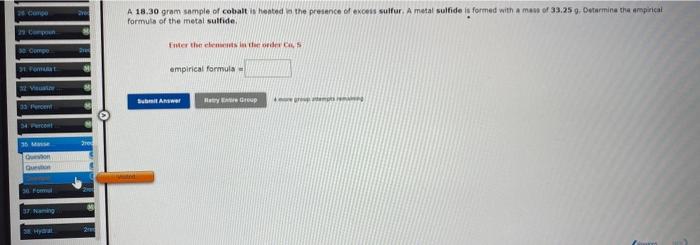
A. 18.30 gram.sample of cobalt is hepted a the preience of exceis suifur. A metal sulfide is formed aith a mass of 33.25 9. Detarmina tha ampinicai formuts of the inetal sulfide. Finer ther eheniants in iber ofiarl Cu, 5 : empirical formula = It is often possible to change a hydrate into an anhydrous compound by heating it to drive off the water (dehydration). A. 39.76 gram sample of a hydrate of MnSO4 was heated thoroughly in a porcelain crucible, until its waight ramained constant. After heating, 23,17 grams of the anhydrous compound remained. What is the formula of the hydrate? tt is aften possible to change a hydrate into an anhydrous compound by heating it to dive of the water (dehydration). A 43.61 gram sample of a hydrate of LiCl was heated tharoughly in s porcelain crucible, untilits meight ramtained constant. After hesting, 30.60 grams of the anhydrous compound remeined. What is the formula of the hydrate? Uie a perioel lentead of a dot in the formoula of the fivitente 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


