Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Calculating mole fraction in a gas mixture 315 A 10.00L tank at 17.5 C is filled with 12.1 g of dinitrogen difluoride gas and 6.44
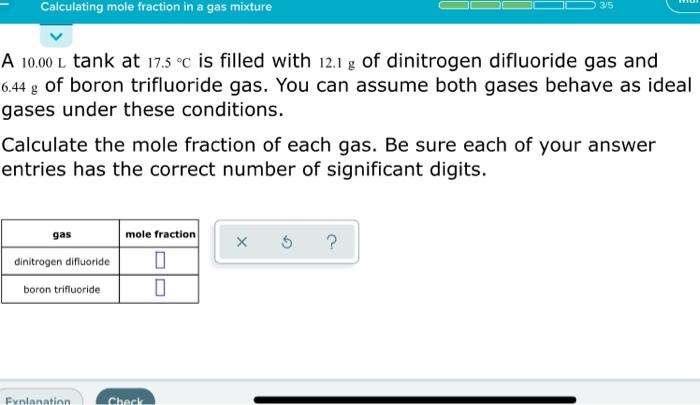 Calculating mole fraction in a gas mixture 315 A 10.00L tank at 17.5 C is filled with 12.1 g of dinitrogen difluoride gas and 6.44 g of boron trifluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Be sure each of your answer entries has the correct number of significant digits. gas X 5 ? dinitrogen difluoride mole fraction 0 0 boron trifluoride Enlanhlor Check
Calculating mole fraction in a gas mixture 315 A 10.00L tank at 17.5 C is filled with 12.1 g of dinitrogen difluoride gas and 6.44 g of boron trifluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction of each gas. Be sure each of your answer entries has the correct number of significant digits. gas X 5 ? dinitrogen difluoride mole fraction 0 0 boron trifluoride Enlanhlor Check

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


