1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia...
Fantastic news! We've Found the answer you've been seeking!
Question:

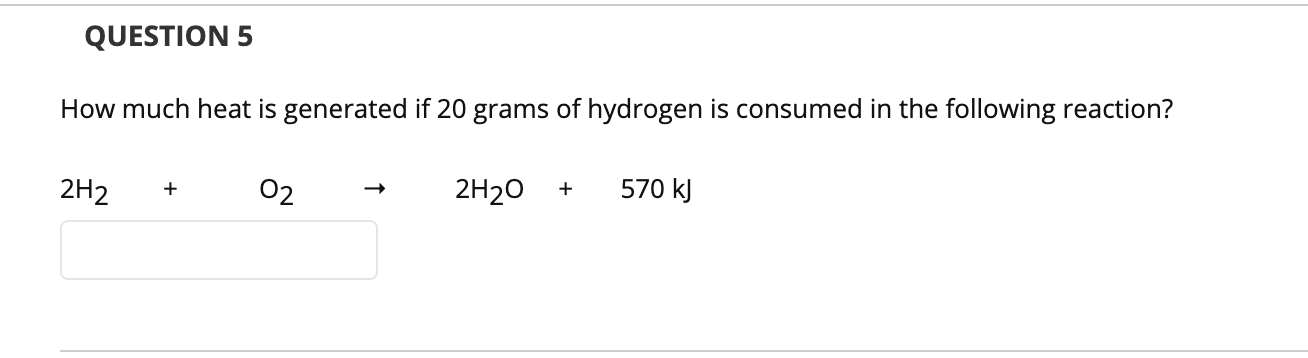
Transcribed Image Text:
1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ 1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Write the net ionic equation for CaCl2(aq) + Pb(NO3)2(aq) ( Ca(NO3)2(aq) + PbCl2(s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Blaine is a practicing dentist. He operates his business from the basement of his house, with a separate entrance and facilities. Blaine uses the simplified method every year to calculate his...
-
Why do governments sometimes impose quantity regulations that limit the level of negative-externality-inducing consumption? Why do governments sometimes impose price regulations by taxing this...
-
Kerry Ellison worked as a revenue agent for the Internal Revenue Service (IRS) in San Mateo, California. Sterling Gray's desk was 20 feet from Ellison's desk, two rows behind and one row over....
-
What is characteristic of a flat structure? A. Narrow span of control. B. Fewer layers in the reporting structure. C. Combines two structures into one. D. Is cost-effective with the use of resources.
-
The intangible assets section of the balance sheet for Vincent Company at December 31, 2012, is presented here. Patents ($70,000 cost less $7,000 amortization) .... $63,000 Copyrights ($48,000 cost...
-
It is common practice for airlines to overbook so that despite any no-shows or cancellations, eachflight will still have as many occupied seats as possible, because an unoccupied seat represents...
-
Based on the information provided in the case, illustrate the pricing game between Sony and Microsoft using a 2-by-2 payoff matrix when Sony and Microsoft charge for their games $399, or lower price...
-
Identify and describe a hypothetical project for use in this course. The project is Office Relocation. The project should consist of work that will take at least 4 months' time, have a beginning and...
-
How do advances in single-cell genomics, transcriptomics, and proteomics technologies revolutionize our understanding of cellular heterogeneity, developmental trajectories, and lineage commitment in...
-
Discuss the evolutionary dynamics and molecular mechanisms driving speciation and diversification in eukaryotic organisms, exploring concepts such as allopatric, sympatric, and parapatric speciation,...
-
What externalities are created by public education? How does the government provide incentives for education, and do you think those incentives are sufficient to achieve the government's goals
-
A company that manufactures cameras has a demand given by the function y=f(x)=-0.2x 2 +60, on the other hand the quantity it is willing to put on the market is given by Y=g(x) =0.1x 2 +x+20. If the...
-
What new ethical issues have arisen in the areas of technology and social work within the last 3 years, especially because of the Covid 19 Pandemic? 2b. How has the NASW Code of Ethics had to adapt...
-
REHCorporation's most recent dividend was $2.54 pershare, its expected annual rate of dividend growth is 5%, and the required return is now 15%. A variety of proposals are being considered by...
-
Propose a reasonable mechanism for the following reaction. OH
-
What is the freezing point of an aqueous solution that boils at 105.0 oC?
-
Specify what ions are present in solution upon dissolving each of the following substances in water: (a) ZnCl2 (b) HNO3 (c) (NH4)2SO4 (d) Ca(OH)2.
-
Detailed calculations show that the value of Zeff for the outermost electrons in Si and Cl atoms is 4.29+ and 6.12+, respectively. (a) What value do you estimate for Zeff experienced by the outermost...
-
Create a statement of cash flow for the Arrow Company for the years ended December 31, 2014 and December 31, 2015 from the balance sheets and income statement created in problem P1.32 from Chapter 1....
-
Recent financial statements for The Hoechst Group, a German conglomerate with operations in agriculture, chemicals, pharmaceuticals, and veterinary products are pre- ver sented as follows: The...
-
The 2015 annual report of The Procter & Gamble Company (P&G) is available at www.pginvestor.com. After reviewing P&Gs annual report, respond to the following questions: a. P&Gs 2015 statement of cash...

Study smarter with the SolutionInn App



