Question
Consider the following stirred-tank mixing process. The control scheme accounts for changes in the concentration CA coming in through stream A by changing the
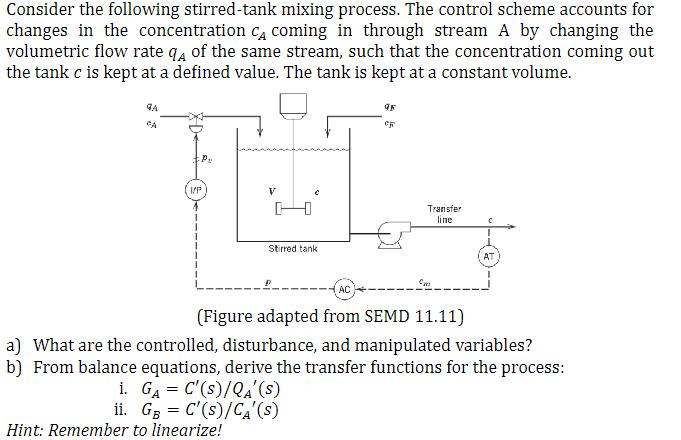
Consider the following stirred-tank mixing process. The control scheme accounts for changes in the concentration CA coming in through stream A by changing the volumetric flow rate 4 of the same stream, such that the concentration coming out the tank c is kept at a defined value. The tank is kept at a constant volume. GA P I/P V Stirred tank Hint: Remember to linearize! i. G = C'(s)/QA' (S) ii. GB = C'(s)/C'(S) AC 45 eg Transfer line Cm C (Figure adapted from SEMD 11.11) a) What are the controlled, disturbance, and manipulated variables? b) From balance equations, derive the transfer functions for the process: AT
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Elements Of Chemical Reaction Engineering
Authors: H. Fogler
6th Edition
013548622X, 978-0135486221
Students also viewed these Chemical Engineering questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



