Answered step by step
Verified Expert Solution
Question
1 Approved Answer
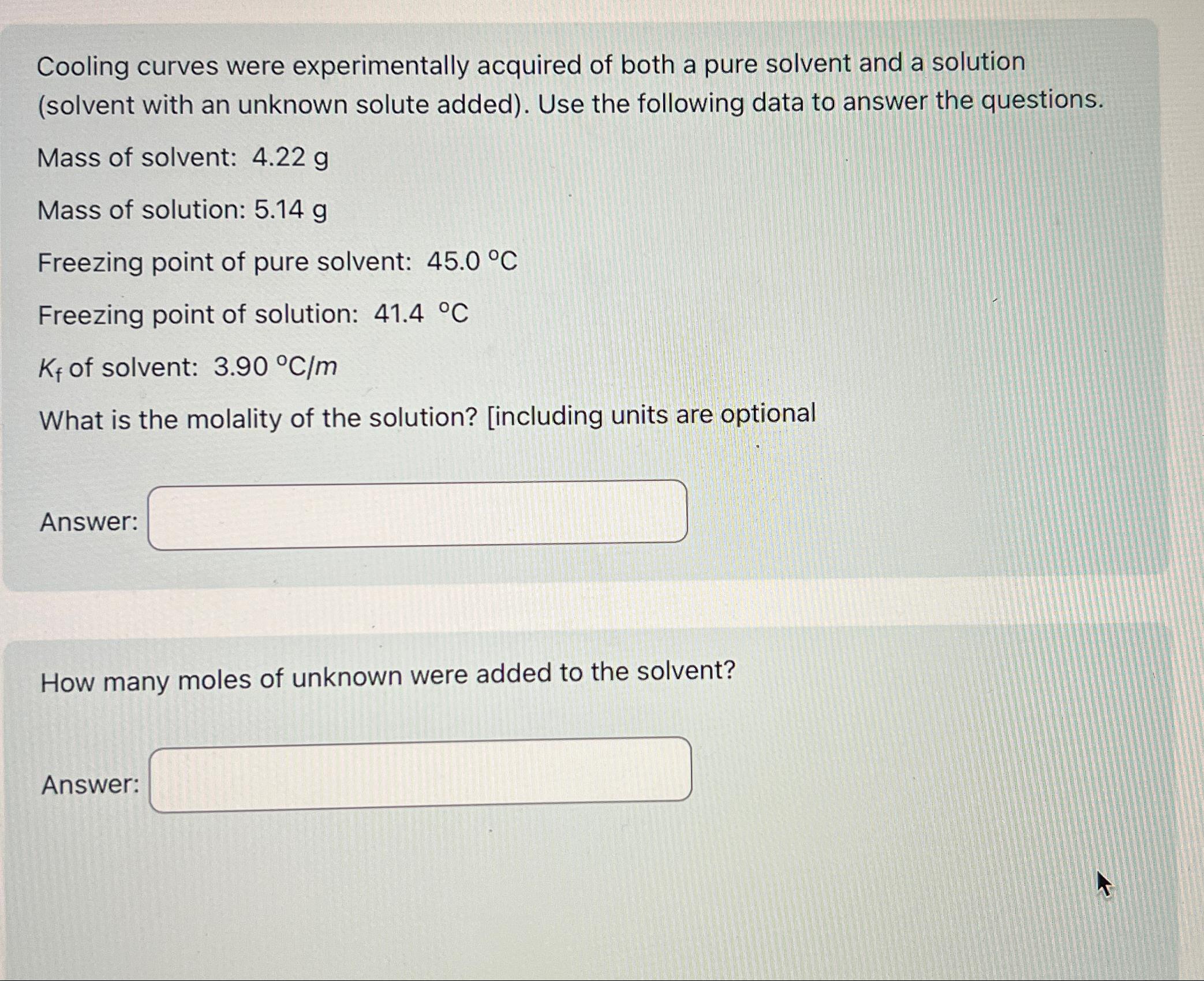
Cooling curves were experimentally acquired of both a pure solvent and a solution (solvent with an unknown solute added). Use the following data to answer
Cooling curves were experimentally acquired of both a pure solvent and a solution (solvent with an unknown solute added). Use the following data to answer the questions.\ Mass of solvent:
4.22g\ Mass of solution:
5.14g\ Freezing point of pure solvent:
45.0\\\\deg C\ Freezing point of solution:
41.\\\\deg C\
K_(f)of solvent:
3.90\\\\deg (C)/(m)\ What is the molality of the solution? [including units are optional\ Answer:\ How many moles of unknown were added to the solvent?\ Answer:
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


