Question
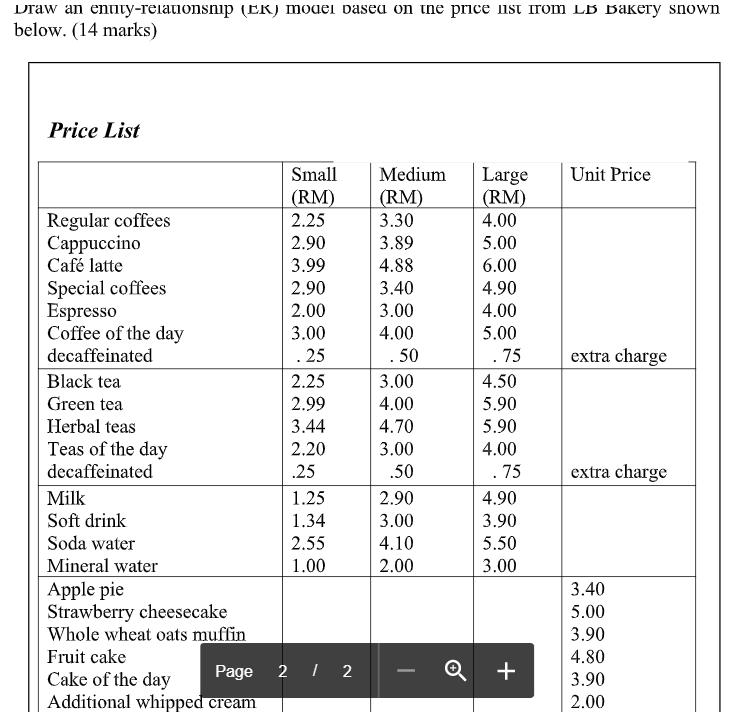
Draw an Entry relationship Draw an enuty-reiaionsnip (ER) moaei basea on ine price ist irom LB Bakery snown below. (14 marks) Price List Small Medium
Draw an Entry relationship
Draw an enuty-reiaionsnip (ER) moaei basea on ine price ist irom LB Bakery snown below. (14 marks) Price List Small Medium Large (RM) Unit Price (RM) (RM) Regular coffees ppuccino Caf latte 2.25 3.30 4.00 2.90 3.89 5.00 3.99 4.88 6.00 Special coffees Espresso Coffee of the day 2.90 3.40 4.90 2.00 3.00 4.00 3.00 4.00 5.00 decaffeinated . 25 . 50 . 75 extra charge Black tea 2.25 3.00 4.50 Green tea 2.99 4.00 5.90 Herbal teas 3.44 4.70 5.90 Teas of the day 2.20 3.00 4.00 decaffeinated .25 .50 . 75 extra charge Milk 1.25 2.90 4.90 Soft drink 1.34 3.00 3.90 Soda water 2.55 4.10 5.50 Mineral water Apple pie Strawberry cheesecake Whole wheat oats muffin Fruit cake 1.00 2.00 3.00 3.40 5.00 3.90 4.80 Page 2 I 2 + Cake of the day Additional whipped cream 3.90 2.00
Step by Step Solution
3.39 Rating (152 Votes )
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Physical Chemistry
Authors: Peter Atkins
7th Edition
978-0716735397, 716735393, 978-0716743880
Students also viewed these Databases questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



