Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Draw the condensed structure and calculate the molar masses (to four significant figures) for each liquid used in this lab. Record your answers in the
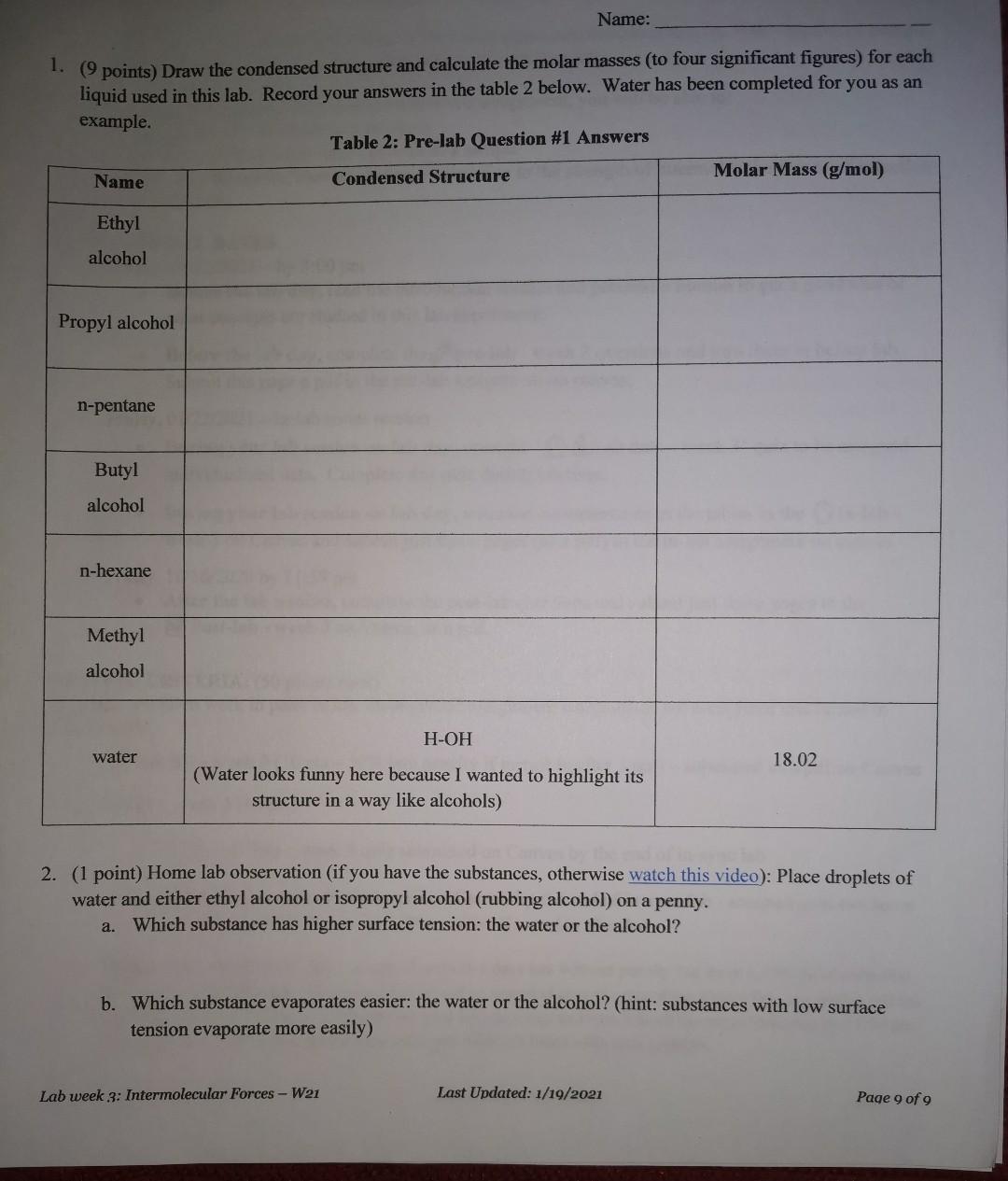
Draw the condensed structure and calculate the molar masses (to four significant figures) for each liquid used in this lab. Record your answers in the table to below. Water has been completed for you as an example.
Name: 1. (9 points) Draw the condensed structure and calculate the molar masses (to four significant figures) for each liquid used in this lab. Record your answers in the table 2 below. Water has been completed for you as an example. Table 2: Pre-lab Question #1 Answers Name Condensed Structure Molar Mass (g/mol) Ethyl alcohol Propyl alcohol n-pentane Butyl alcohol n-hexane Methyl alcohol H-OH water 18.02 (Water looks funny here because I wanted to highlight its structure in a way like alcohols) 2. (1 point) Home lab observation (if you have the substances, otherwise watch this video): Place droplets of water and either ethyl alcohol or isopropyl alcohol (rubbing alcohol) on a penny. a. Which substance has higher surface tension: the water or the alcohol? b. Which substance evaporates easier the water or the alcohol? (hint: substances with low surface tension evaporate more easily) Lab week 3: Intermolecular Forces - W21 Last Updated: 1/19/2021 Page 9 of 9Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


