Answered step by step
Verified Expert Solution
Question
1 Approved Answer
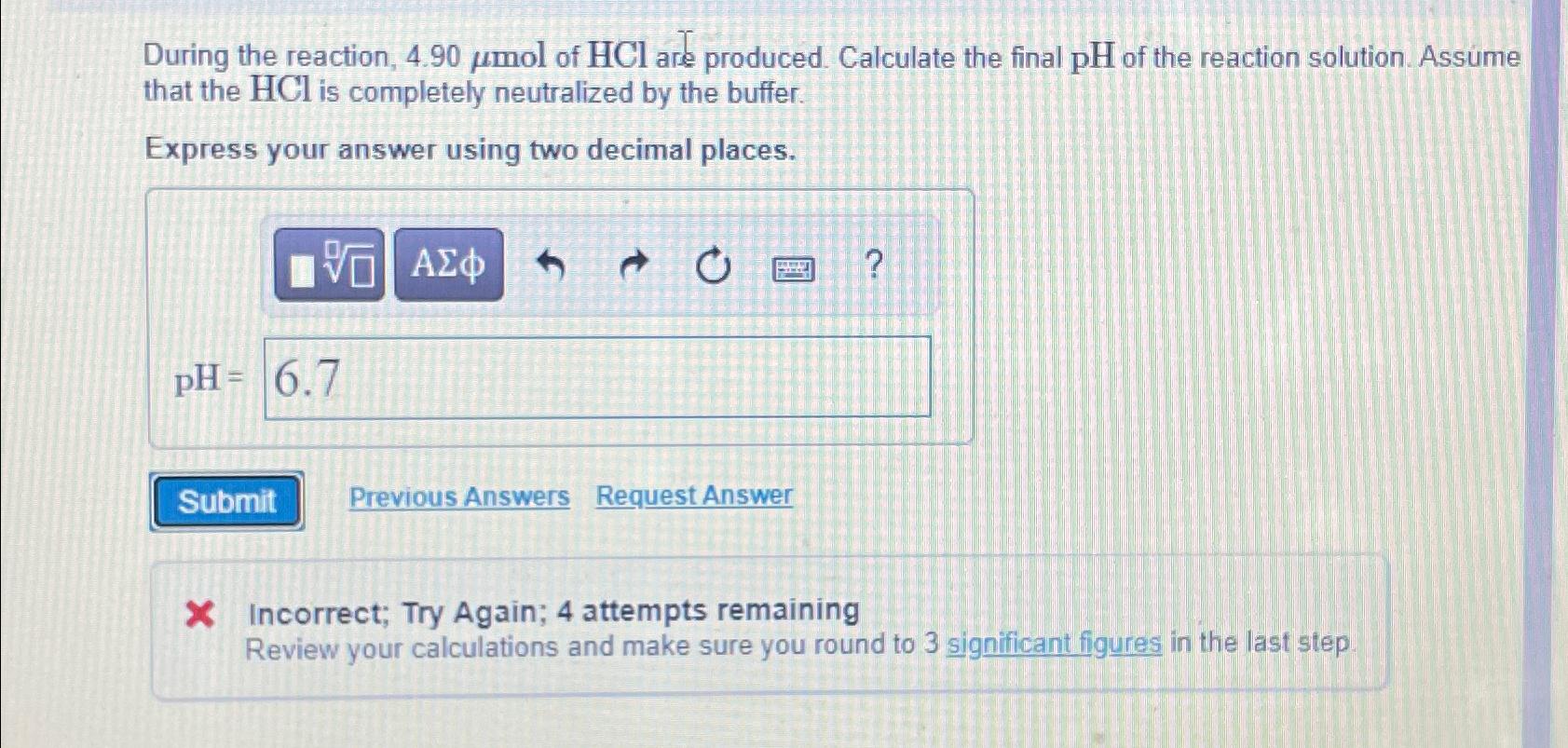
During the reaction, 4 . 9 0 m o l of H C l are produced. Calculate the final p H of the reaction solution.
During the reaction, of are produced. Calculate the final of the reaction solution. Assume that the is completely neutralized by the buffer.
Express your answer using two decimal places.
Previous Answers
X Incorrect; Try Again; attempts remaining
Review your calculations and make sure you round to significant figures in the last step.
A biochemical reaction takes place in a solution of phosphate buffer initially at
tableAcid Proton DonorConjugate Base Proton Acc,,
Please help!
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


