Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Elementary irreversible gas phase reaction is carried out in a catalyst filled piston flow adiabatic reactor. Pure substance A is fed into the reactor at
Elementary irreversible gas phase reaction is carried out in a catalyst filled piston flow adiabatic reactor. Pure substance A is fed into the reactor at a temperature of 450 K and a pressure of 10 atm, at a volumetric flow rate of 20 dm3/s. Using the data below;
Elementary irreversible gas phase reaction is carried out in a catalyst filled piston flow adiabatic reactor. Pure substance A is fed into the reactor at a temperature of 450 K and a pressure of 10 atm, at a volumetric flow rate of 20 dm3/s. Using the data below;
a) Construct the stoichiometric table and write the reaction rate in terms of conversion.
b) Draw iso-rate curves for -r'''= 0.004 and -r''' = 0.075 (mol/g cat.s)
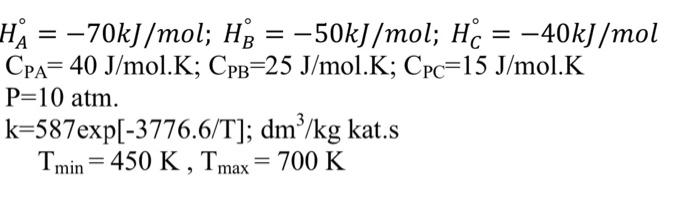
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


