Answered step by step
Verified Expert Solution
Question
1 Approved Answer
fill out the cart with current information [Cu2+] in stock solution- 0.22M [Cu2+] in Solution 1-0.022M [Cu2+] in M: Solution 2-0.0044 [Cu2+] in M: Solution
fill out the cart with current information
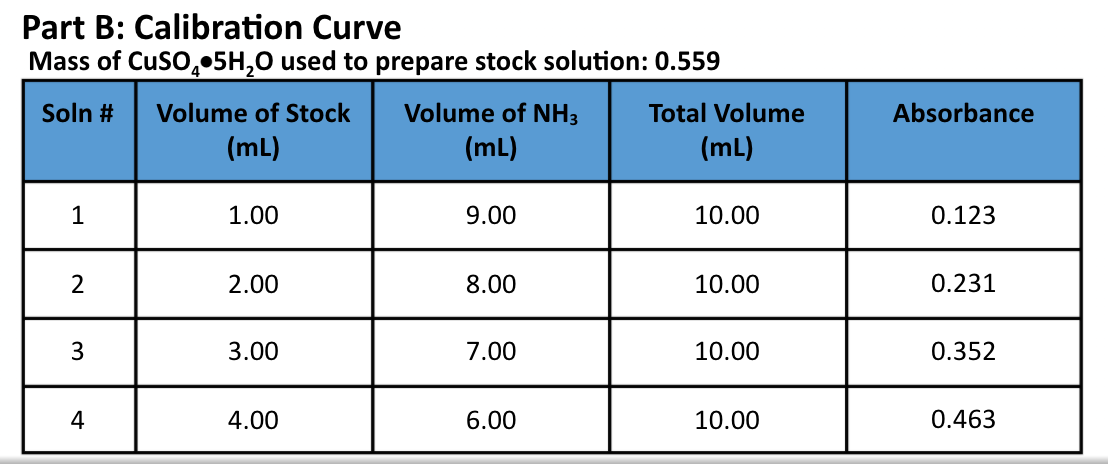
[Cu2+] in stock solution- 0.22M
[Cu2+] in Solution 1-0.022M
[Cu2+] in M: Solution 2-0.0044
[Cu2+] in M: Solution 3-0.0066
[Cu2+] in M: Solution 4-0.0088
PART C
beaker 1- mass of complex -0.100g
beaker 2 -mass of complex -0.127g
beaker 3 - mass of complex 0.134g
0.5M NH3 to 2nd beaker distilled water to the first beaker.
absorbance compelx 1 - 0.141
absorbance compelx 2- 0.157
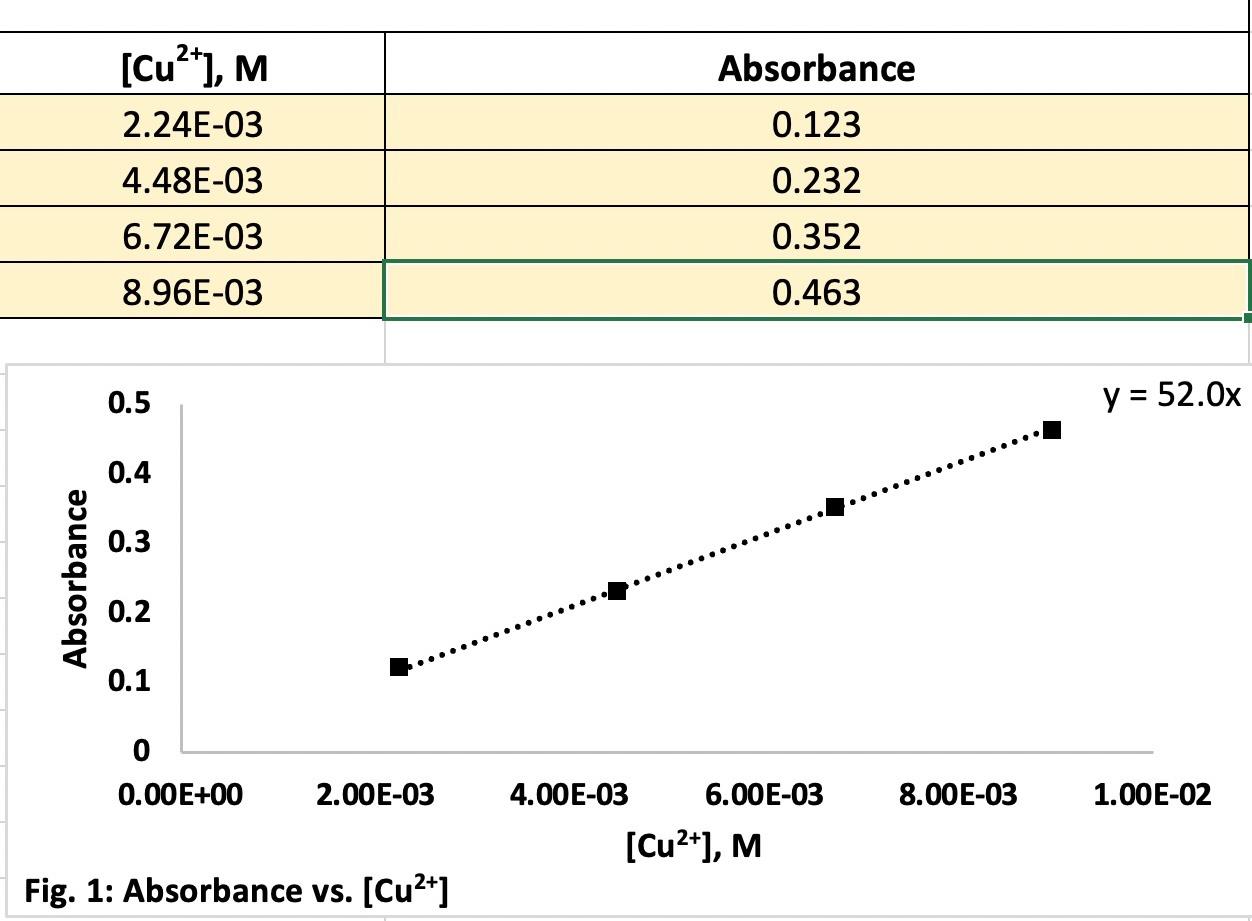
equation from the calibration curve - y=52.0x
question - fill in the data table

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


