Answered step by step
Verified Expert Solution
Question
1 Approved Answer
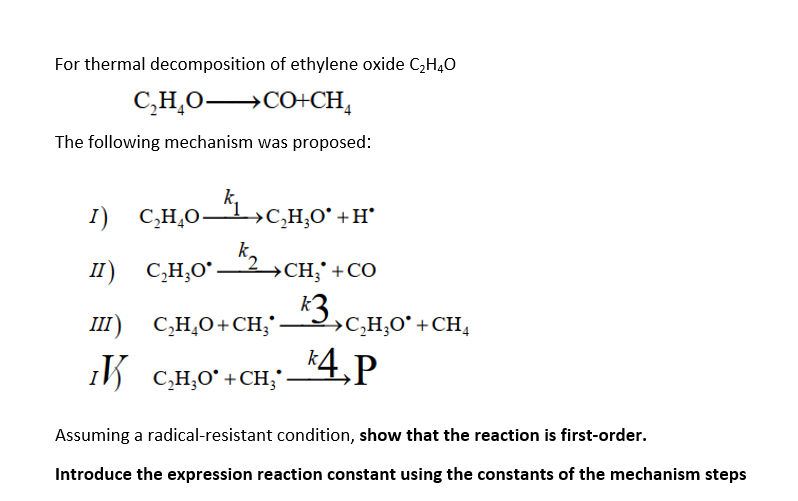
For thermal decomposition of ethylene oxide C2H40 C,H,O+CO+CH, The following mechanism was proposed: k 1) C,H0_51,c.40*+H *1,0,1,0 * II) C,H,09 2ych; CH;' +CO III) C,H,O+CH;
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


