Answered step by step
Verified Expert Solution
Question
1 Approved Answer
how many moles of HF are needed to react with 0.300 mol of Na2SiO3 several brands of antacids use AlOH3 to react with stomic acid
how many moles of HF are needed to react with 0.300 mol of Na2SiO3 several brands of antacids use AlOH3 to react with stomic acid which contains primarily hcl calculate the number of germs of hcl that can react with .500 g of AlOH3 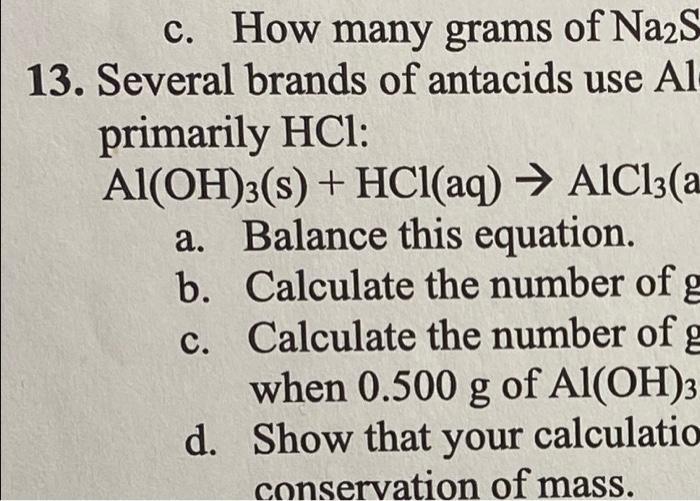 c. How many grams of Na2S 13. Several brands of antacids use Al primarily HCl : Al(OH)3(s)+HCl(aq)AlCl3(a a. Balance this equation. b. Calculate the number of g c. Calculate the number of g when 0.500g of Al(OH)3 d. Show that your calculatio conservation of mass
c. How many grams of Na2S 13. Several brands of antacids use Al primarily HCl : Al(OH)3(s)+HCl(aq)AlCl3(a a. Balance this equation. b. Calculate the number of g c. Calculate the number of g when 0.500g of Al(OH)3 d. Show that your calculatio conservation of mass
how many moles of HF are needed to react with 0.300 mol of Na2SiO3
several brands of antacids use AlOH3 to react with stomic acid which contains primarily hcl calculate the number of germs of hcl that can react with .500 g of AlOH3
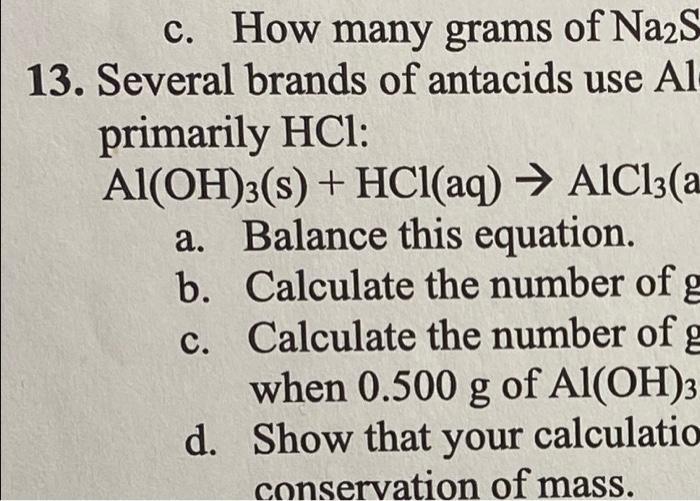
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


