I need help figuring out these problems. if you could please help that would be lovely and will you please write clearly. Thank you
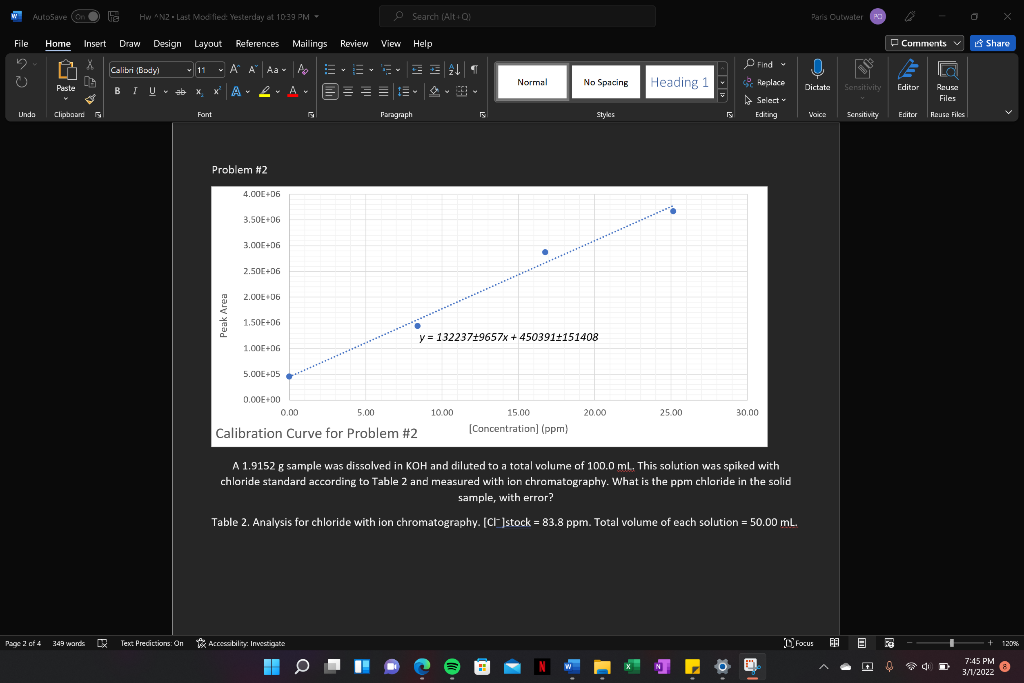
Problem #2
A 1.9152 g sample was dissolved in KOH and diluted to a total volume of 100.0 mL. This solution was spiked with chloride standard according to Table 2 and measured with ion chromatography. What is the ppm chloride in the solid sample, with error?
Table 2. Analysis for chloride with ion chromatography. [Cl ]stock = 83.8 ppm. Total volume of each solution = 50.00 mL.
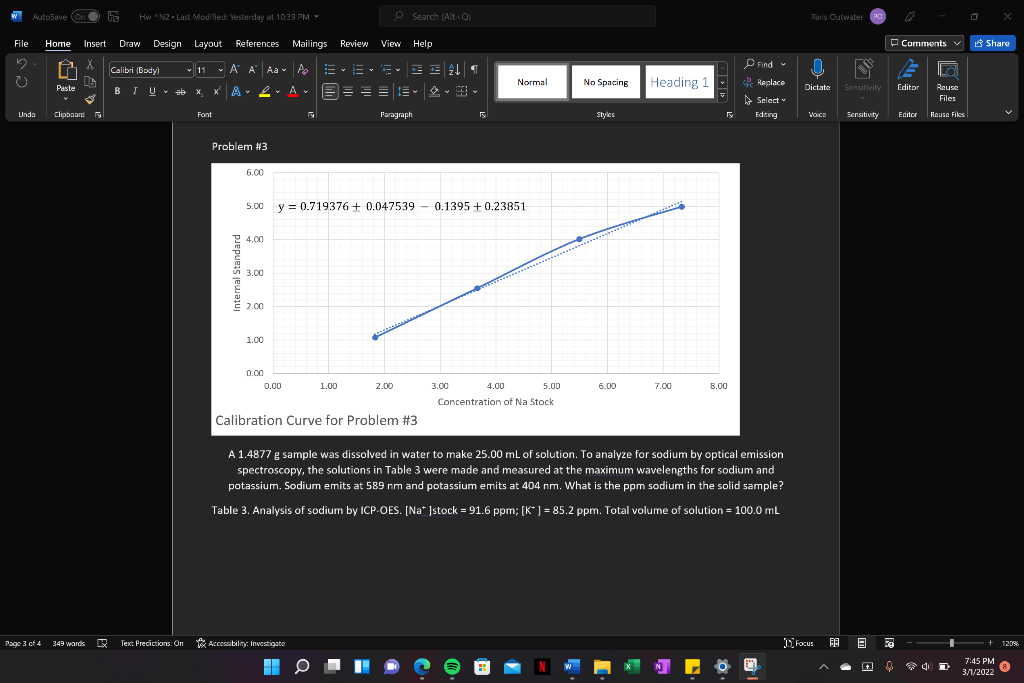
Problem #3
A 1.4877 g sample was dissolved in water to make 25.00 mL of solution. To analyze for sodium by optical emission spectroscopy, the solutions in Table 3 were made and measured at the maximum wavelengths for sodium and potassium. Sodium emits at 589 nm and potassium emits at 404 nm. What is the ppm sodium in the solid sample?
Table 3. Analysis of sodium by ICP-OES. [Na+ ]stock = 91.6 ppm; [K+ ] = 85.2 ppm. Total volume of solution = 100.0 mL
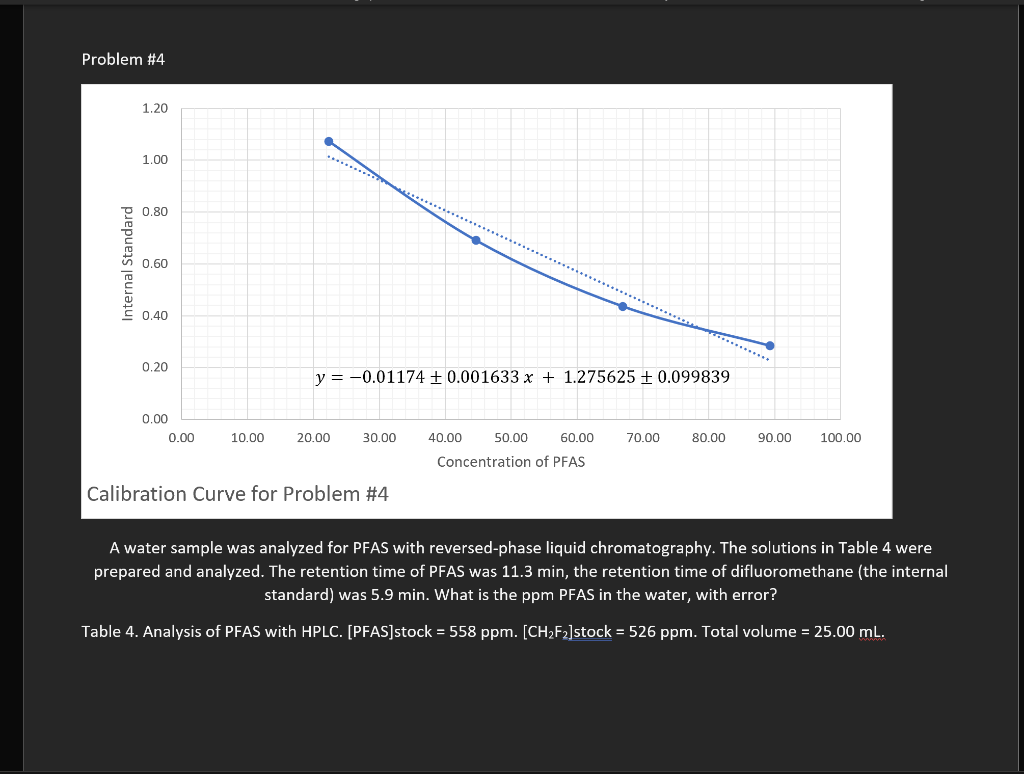
Problem #4
A water sample was analyzed for PFAS with reversed-phase liquid chromatography. The solutions in Table 4 were prepared and analyzed. The retention time of PFAS was 11.3 min, the retention time of difluoromethane (the internal standard) was 5.9 min. What is the ppm PFAS in the water, with error?
Table 4. Analysis of PFAS with HPLC. [PFAS]stock = 558 ppm. [CH2F2]stock = 526 ppm. Total volume = 25.00 mL.
AutoSave (on HwN2 - Last Modified: Yesterday at 10:39 PM - Search (Alto Paris Outwater File Home Insert Draw Design Layout References Mailings Review View Help Comments Share Find O Calibri (body) 11 A A Aa A EE # 21 = BIU * X *A 2 ab x A EEEE - Normal No Spacing Replace Heading 1 U Dictate Paste Sensitivity Editor Reuse Files Select Unda Clipboard Font Paragraph Styles 2 Editing VOICE Sensitivity Editor Rouse Files Problem #2 4.00E+06 3.50E+06 3.00E-06 2.50E+06 2.00E+06 Peak Area 1.50E+06 y = 13223719657x + 450391151408 1.00E+OG 5.00E+05 0.00E+00 0.00 5.00 10.00 20.00 25.00 30.00 15.00 [Concentration (ppm) Calibration Curve for Problem #2 A 1.9152 g sample was dissolved in KOH and diluted to a total volume of 100.0 ml. This solution was spiked with chloride standard according to Table 2 and measured with ion chromatography. What is the ppm chloride in the solid sample, with error? Table 2. Analysis for chloride with ion chromatography. [C]stock = 83.8 ppm. Total volume of each solution = 50.00 mL. Page 2 of 4 349 wands DY Text Predictions: On Accessibility: Investigate Focus E + 120% 0 7:45 PM 3/1//2022 AutoSave (on HwN2 - Last Modified: Yesterday at 10:39 PM - Search (Alt ! Paris Outwater File Home Insert Draw Design Layout References Mailings Review View Help Comments v Share Find O Calibri (body) 11 A A A A E ! BIU * X * A 2 ab x A EE EE 9 1 Normal No Spacing Heading 1 Replace U Dictate Paste Sensitivity Editor Reuse Files Select Editing Unda Clipboard Font Paragraph Styles 2 Voice Sensitivity Editor Rouse Files Problem #3 6.00 5.00 y = 0.719376 + 0.047539 0.1395 + 0.23851 24.00 Internal Standard 3.00 2.00 1.00 0.00 0.00 1.00 2.00 6.00 7.00 8.00 3.00 4.00 5.00 Concentration of Na Stock Calibration Curve for Problem #3 A A 1.4877 g sample was dissolved in water to make 25.00 mL of solution. To analyze for sodium by optical emission spectroscopy, the solutions in Table 3 were made and measured at the maximum wavelengths for sodium and potassium. Sodium emits at 589 nm and potassium emits at 404 nm. What is the ppm sodium in the solid sample? Table 3. Analysis of sodium by ICP-OES. [Najstock = 91.6 ppm; [K* ] = 85.2 ppm. Total volume of solution - 100.0 mL Page 04 349 wands DR Text Predictions: On Accessibility: Investigate Focus E + 120% O 7:45 PM 3/1//2022 Problem #4 1.20 1.00 D 0.80 Internal Standard 0.60 S 0.40 0.20 y = -0.01174 + 0.001633 x + 1.275625 + 0.099839 0.00 0.00 10.00 20.00 30.00 40.00 50.00 60.00 70.00 80.00 90.00 100.00 Concentration of PFAS Calibration Curve for Problem #4 A water sample was analyzed for PFAS with reversed-phase liquid chromatography. The solutions in Table 4 were prepared and analyzed. The retention time of PFAS was 11.3 min, the retention time of difluoromethane (the internal standard) was 5.9 min. What is the ppm PFAS in the water, with error? Table 4. Analysis of PFAS with HPLC. (PFAS]stock = 558 ppm. [CH2F2]stock = 526 ppm. Total volume = 25.00 ml. AutoSave (on HwN2 - Last Modified: Yesterday at 10:39 PM - Search (Alto Paris Outwater File Home Insert Draw Design Layout References Mailings Review View Help Comments Share Find O Calibri (body) 11 A A Aa A EE # 21 = BIU * X *A 2 ab x A EEEE - Normal No Spacing Replace Heading 1 U Dictate Paste Sensitivity Editor Reuse Files Select Unda Clipboard Font Paragraph Styles 2 Editing VOICE Sensitivity Editor Rouse Files Problem #2 4.00E+06 3.50E+06 3.00E-06 2.50E+06 2.00E+06 Peak Area 1.50E+06 y = 13223719657x + 450391151408 1.00E+OG 5.00E+05 0.00E+00 0.00 5.00 10.00 20.00 25.00 30.00 15.00 [Concentration (ppm) Calibration Curve for Problem #2 A 1.9152 g sample was dissolved in KOH and diluted to a total volume of 100.0 ml. This solution was spiked with chloride standard according to Table 2 and measured with ion chromatography. What is the ppm chloride in the solid sample, with error? Table 2. Analysis for chloride with ion chromatography. [C]stock = 83.8 ppm. Total volume of each solution = 50.00 mL. Page 2 of 4 349 wands DY Text Predictions: On Accessibility: Investigate Focus E + 120% 0 7:45 PM 3/1//2022 AutoSave (on HwN2 - Last Modified: Yesterday at 10:39 PM - Search (Alt ! Paris Outwater File Home Insert Draw Design Layout References Mailings Review View Help Comments v Share Find O Calibri (body) 11 A A A A E ! BIU * X * A 2 ab x A EE EE 9 1 Normal No Spacing Heading 1 Replace U Dictate Paste Sensitivity Editor Reuse Files Select Editing Unda Clipboard Font Paragraph Styles 2 Voice Sensitivity Editor Rouse Files Problem #3 6.00 5.00 y = 0.719376 + 0.047539 0.1395 + 0.23851 24.00 Internal Standard 3.00 2.00 1.00 0.00 0.00 1.00 2.00 6.00 7.00 8.00 3.00 4.00 5.00 Concentration of Na Stock Calibration Curve for Problem #3 A A 1.4877 g sample was dissolved in water to make 25.00 mL of solution. To analyze for sodium by optical emission spectroscopy, the solutions in Table 3 were made and measured at the maximum wavelengths for sodium and potassium. Sodium emits at 589 nm and potassium emits at 404 nm. What is the ppm sodium in the solid sample? Table 3. Analysis of sodium by ICP-OES. [Najstock = 91.6 ppm; [K* ] = 85.2 ppm. Total volume of solution - 100.0 mL Page 04 349 wands DR Text Predictions: On Accessibility: Investigate Focus E + 120% O 7:45 PM 3/1//2022 Problem #4 1.20 1.00 D 0.80 Internal Standard 0.60 S 0.40 0.20 y = -0.01174 + 0.001633 x + 1.275625 + 0.099839 0.00 0.00 10.00 20.00 30.00 40.00 50.00 60.00 70.00 80.00 90.00 100.00 Concentration of PFAS Calibration Curve for Problem #4 A water sample was analyzed for PFAS with reversed-phase liquid chromatography. The solutions in Table 4 were prepared and analyzed. The retention time of PFAS was 11.3 min, the retention time of difluoromethane (the internal standard) was 5.9 min. What is the ppm PFAS in the water, with error? Table 4. Analysis of PFAS with HPLC. (PFAS]stock = 558 ppm. [CH2F2]stock = 526 ppm. Total volume = 25.00 ml









