Answered step by step
Verified Expert Solution
Question
1 Approved Answer
I will upload two times pls answers one time to compare answer Take lest. IA #2. Priysical properties Ol Alkalles, AICOHOIS and curiers QUESTION 1

I will upload two times pls answers one time to compare answer
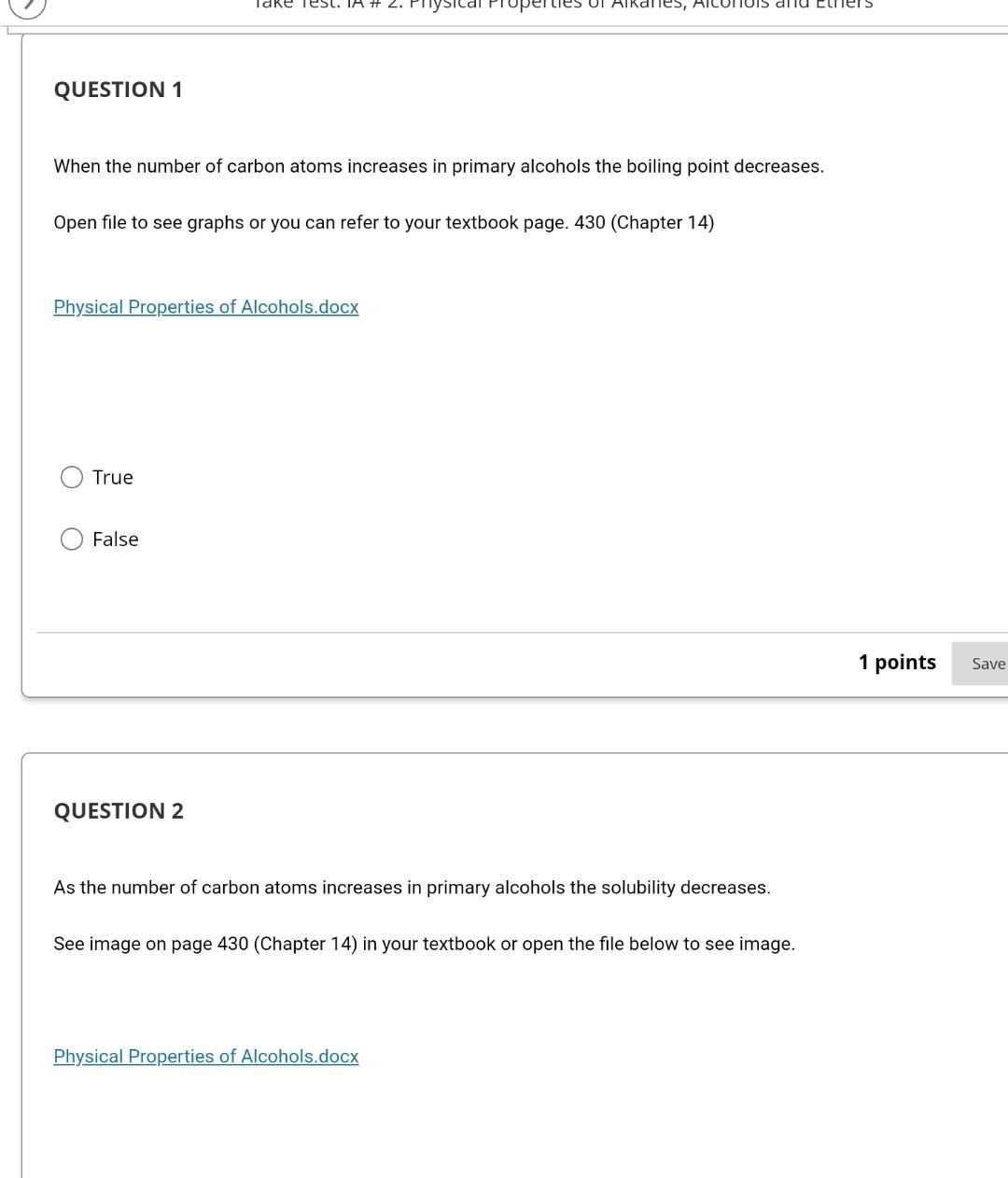
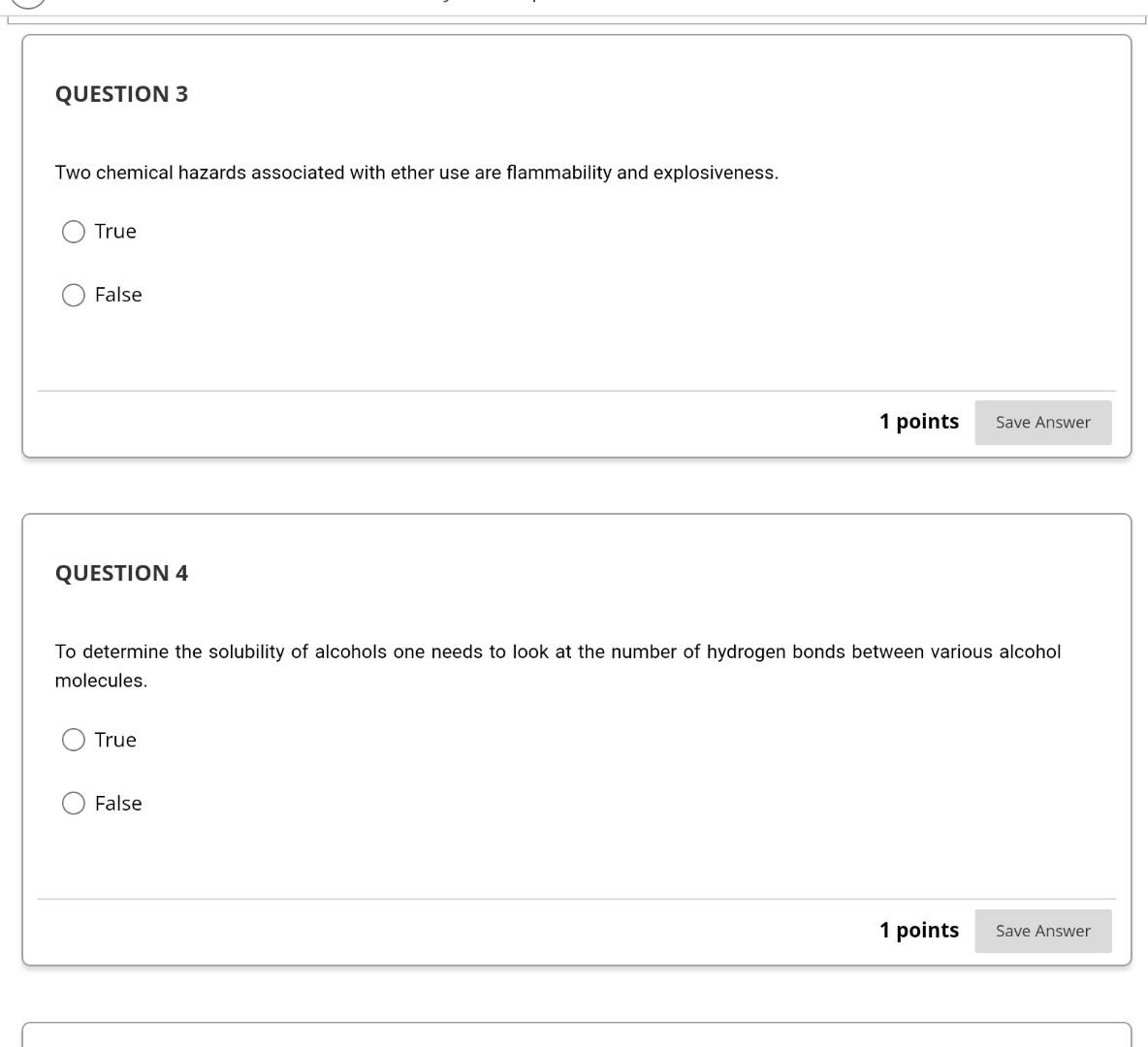
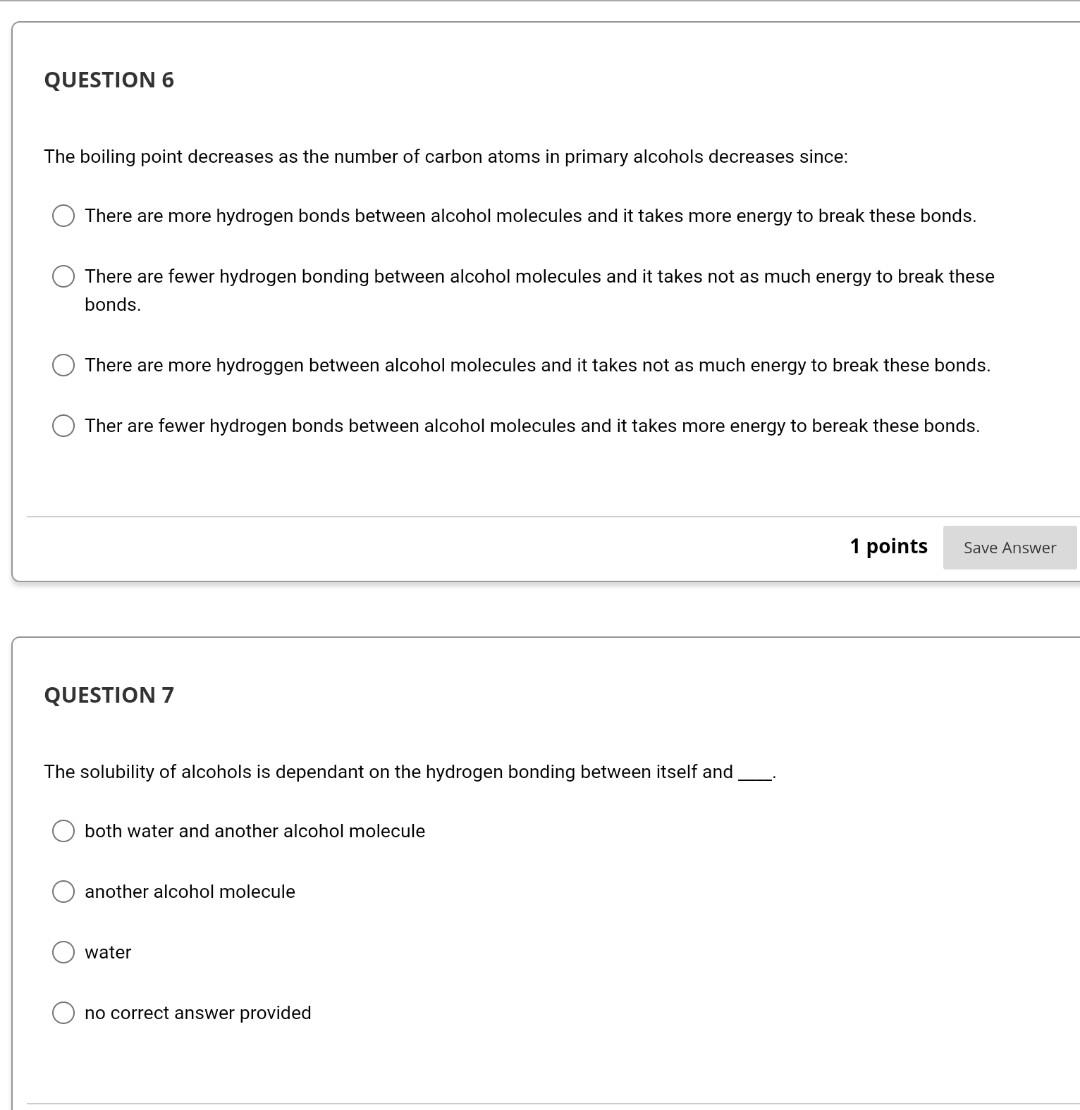
Take lest. IA #2. Priysical properties Ol Alkalles, AICOHOIS and curiers QUESTION 1 When the number of carbon atoms increases in primary alcohols the boiling point decreases. Open file to see graphs or you can refer to your textbook page. 430 (Chapter 14) Physical Properties of Alcohols.docx True False 1 points Save QUESTION 2 As the number of carbon atoms increases in primary alcohols the solubility decreases, See image on page 430 (Chapter 14) in your textbook or open the file below to see image. Physical Properties of Alcohols.docx QUESTION 3 Two chemical hazards associated with ether use are flammability and explosiveness. True False 1 points Save Answer QUESTION 4 To determine the solubility of alcohols one needs to look at the number of hydrogen bonds between various alcohol molecules. True False 1 points Save Answer QUESTION 6 The boiling point decreases as the number of carbon atoms in primary alcohols decreases since: There are more hydrogen bonds between alcohol molecules and it takes more energy to break these bonds. There are fewer hydrogen bonding between alcohol molecules and it takes not as much energy to break these bonds. There are more hydroggen between alcohol molecules and it takes not as much energy to break these bonds. Ther are fewer hydrogen bonds between alcohol molecules and it takes more energy to bereak these bonds. 1 points Save Answer QUESTION 7 The solubility of alcohols is dependant on the hydrogen bonding between itself and both water and another alcohol molecule another alcohol molecule water no correct answer provided QUESTION 8 As the number of carbon atoms increase in primary alcohols the solubility of primary alcohol decreases since: The increased nonpolar carbon length dominates over the polar hydroxyl group. O The increased polar carbon length dominates over the nonpolar hydroxyl group. The increased nonpolar carbon length dominates over the nonpolar hydroxyl group. The increased polar carbon length dominates over the polar hydroxyl group. O 1 points Save Answer QUESTION 9 group of one When drawing hydrogen bonding between alcohol molecules the of the oxygen atom in the alcohol molecule forms a hydrogen bond with another alcohol molecule's in the group. hydroxyl group, oxygen, lone pair of electrons, hydroxyl group lone pair of electrons, hydrogen, lone pair of electrons, lone pair of electrons O hydroxyl group, oxygen, hydrogen, hydroxyl group O lone pair of electrons, hydroxyl group, hydrogen, hydroxyl groupStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


