Answered step by step
Verified Expert Solution
Question
1 Approved Answer
ICE tables are used for calculating changes in concentration in an equilibrium system. I represents the initial concentration, C the change in concentration between the

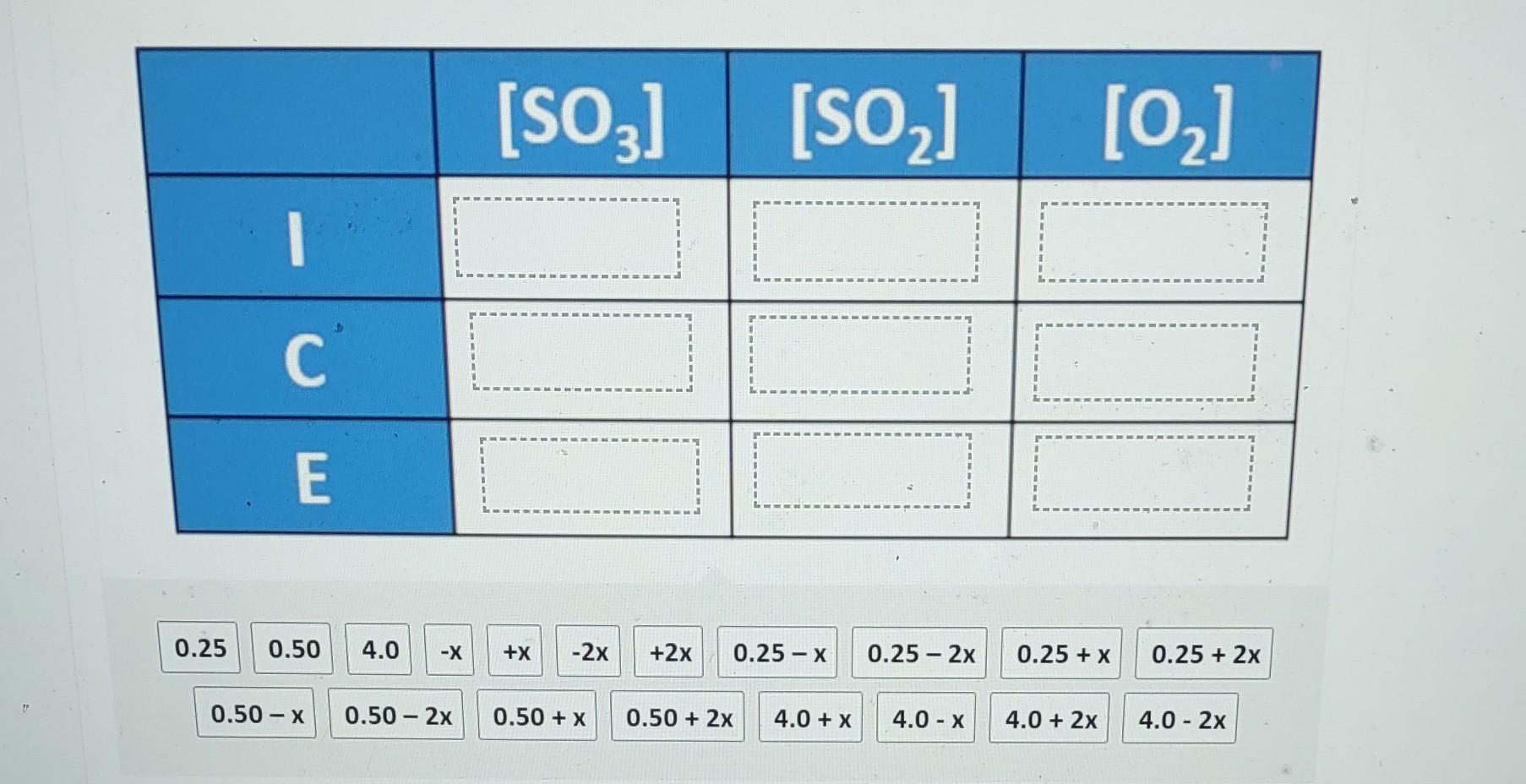
ICE tables are used for calculating changes in concentration in an equilibrium system. I represents the initial concentration, C the change in concentration between the initial value and the equilibrium value, and E the final concentration at equilibrium. The equilibrium constant for the following system is 0.0075 at 373K. 2SO3(g)2SO2(g)+O2(g) The initial concentrations of SO3,SO2, and O2 are 4.0M,0.25M, and 0.50M respectively. Complete the ICE table to show the changes and equilibrium concentrations using the values from the following list. \begin{tabular}{|c|c|c|c|} \hline & {[SO3]} & {[SO2]} & {[O2]} \\ \hline I & & & \\ \hline C & & & \\ \hline E & & & \\ \hline \end{tabular} 0.50x0.502x0.50+x0.50+2x4.0+x4.0x4.0+2x4.02x
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


