Answered step by step
Verified Expert Solution
Question
1 Approved Answer
In a CSTR reactor the following irreversible elementary reaction occurs A + B --> Products where the consumption rate of A and B is given
In a CSTR reactor the following irreversible elementary reaction occurs A + B --> Products
where the consumption rate of A and B is given by:
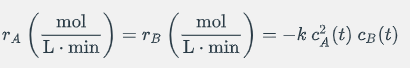
The content of the reactor is perfectly mixed and the temperature, volume and density of the reaction mixture can be assumed constant. Assume that the concentrations of reactant A and B in the feed are CAin= 3.5[mol/L] and CBin= 8[mol/L], respectively.
* Obtain the transfer functions that relate the outlet concentrations, CA(s) and CB(s), to the changes in the inlet flows, FA(s) and FB(s) [L/min]. That is, obtain the 4 possible transfer functions of the system. * Considering CAeq= 5CBeq, where CBeq= 4 mol/L, and a flow rate q= 10 L/min, for a reactor volume of 65 liters and a reaction rate k= 3 L^2/(mol^2*min); Obtain the transfer function CA(s) with respect to the input molar flow FA(s). Also obtain the transfer function CB(s)/FB(s). * Show a comparison of the transfer function models with respect to the numerical solution and comment on the result of the linear approximation with respect to the coefficient of determination R^2 in 35 minutes of operation. * Deduce the value of the damping factor, natural frequency and gain of each of the transfer functions. * Is this second-order system underdamped, critically damped, or overdamped?TA mol Lmin =TB mol L. min In ) ---- cz(t) cp (t) :-k t
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


