Question
Mass Transport Air containing an undesired substance A is to be treated in a wetted-wall column, as shown in Fig.. The air inside the column
Mass Transport
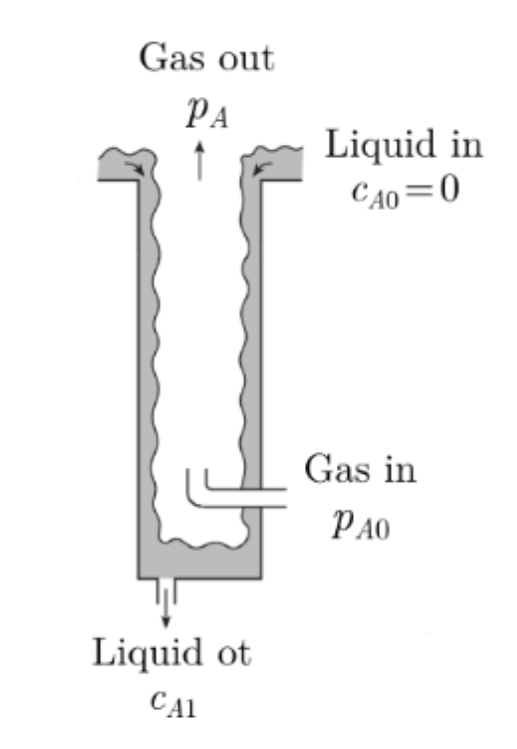
Air containing an undesired substance A is to be treated in a wetted-wall column, as shown in Fig.. The air inside the column contains A at a partial pressure pA= 20 Pa. The air is well mixed right up to the water-air interface. The water film has a thickness of 0.07 cm and flows with an average water velocity of 3 cm/s. The flow in the water-film is laminar and transport into the film is by diffusion only. The column diameter is 10 cm and its length is 20 cm. The diffusion coefficient of A in water is 1.8 109 m2/s and the solubility of A in water is 6.2 Pa m3/mol.
(a) Calculate the concentration of A at the liquid side of the gas- liquid interface. (b) Use an adequate Sherwood correlation to calculate the mass transfer coefficient for the transfer of A from the gas to the bulk of the water film. (c) Calculate the concentration of the water leaving the column.
Tip/ Use mass balanse: in - out = accumilation
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


