Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Need help with 3, 6,7, 8 and 9 Directions: For the electron configuration 1 is rendas 12 Write your selentific notation with two decimal points.
Need help with 3, 6,7, 8 and 9 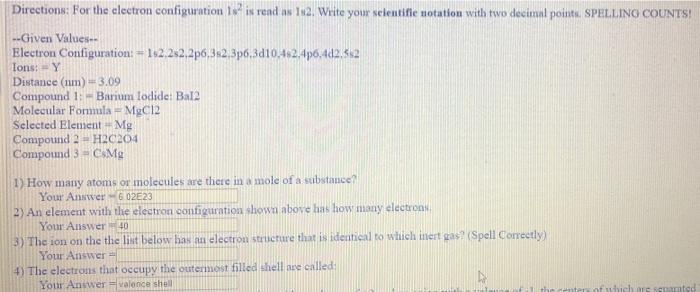

Directions: For the electron configuration 1 is rendas 12 Write your selentific notation with two decimal points. SPELLING COUNTS! --Given Values Electron Configuration: = 152,252.2p6, 3:2,3p6,3d10,482.4p6,402552 Ions: -Y Distance (num) -3,09 Compound 1: Barium Iodide Ba12 Molecular Formula = MgCl2 Selected Element - Mg Compound 2 - H20204 Compound 3.-C.M 1) How many atoms or molecules are there in a mole of a substance Your Answer-602E23 2) An element with the electron contiguration shown above has how many electron Your Answer 40 3) The ion on the the list below has an electron structure that is identical to which inert gas (Spell Correctly) Your Answer 4) The electrons that occupy the outermost filled shell are called Your Answer - Valence shell of which are mod 1) How many atoms or molecules are there in a mole of a substance? Your Answer 6.02E23 2) An element with the electron configuration shown above luns how many electrons, Your Answer 40 3) The son on the the list below has an electron structure that is identical to which inert pax (Spell Correctly) Your Answer 4) The electrons that occupy the outermosttilledshell are called Your AnswerValence the 5) Calculate the force of attraction between nation with a valence of +2 and an anion with valence of the center of which are separated by a distance in um shown above: Your Anwwer-4.62E 11 6) Compute the percent ionic character of the interatomic bonds for Compound I found above Your Answer 7) From the Molecular Fondula given above, what is the percentage (by weight) of the selected clement: Your Answer 8) For Compound 2. indicate the type of primary interatomic) bonding (Tonic, Covalent, or Metallie) Your Anwee 9) For Compound 3. indicate the type of primary interatomic) bonding (Ionic Corulento Metallic Your 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


