Question: Note: Remember that valid rate expressions (revisit Foglers Chapter 10) can only be a function of the concentrations of all relevant species (gas-phase, NOT surface
Note: Remember that valid rate expressions (revisit Foglers Chapter 10) can only be a function of the concentrations of all relevant species (gas-phase, NOT surface concentrations), combinations of rate constants, and Ct, meaning, the total number of active sites (which can generally be titrated by chemisorption).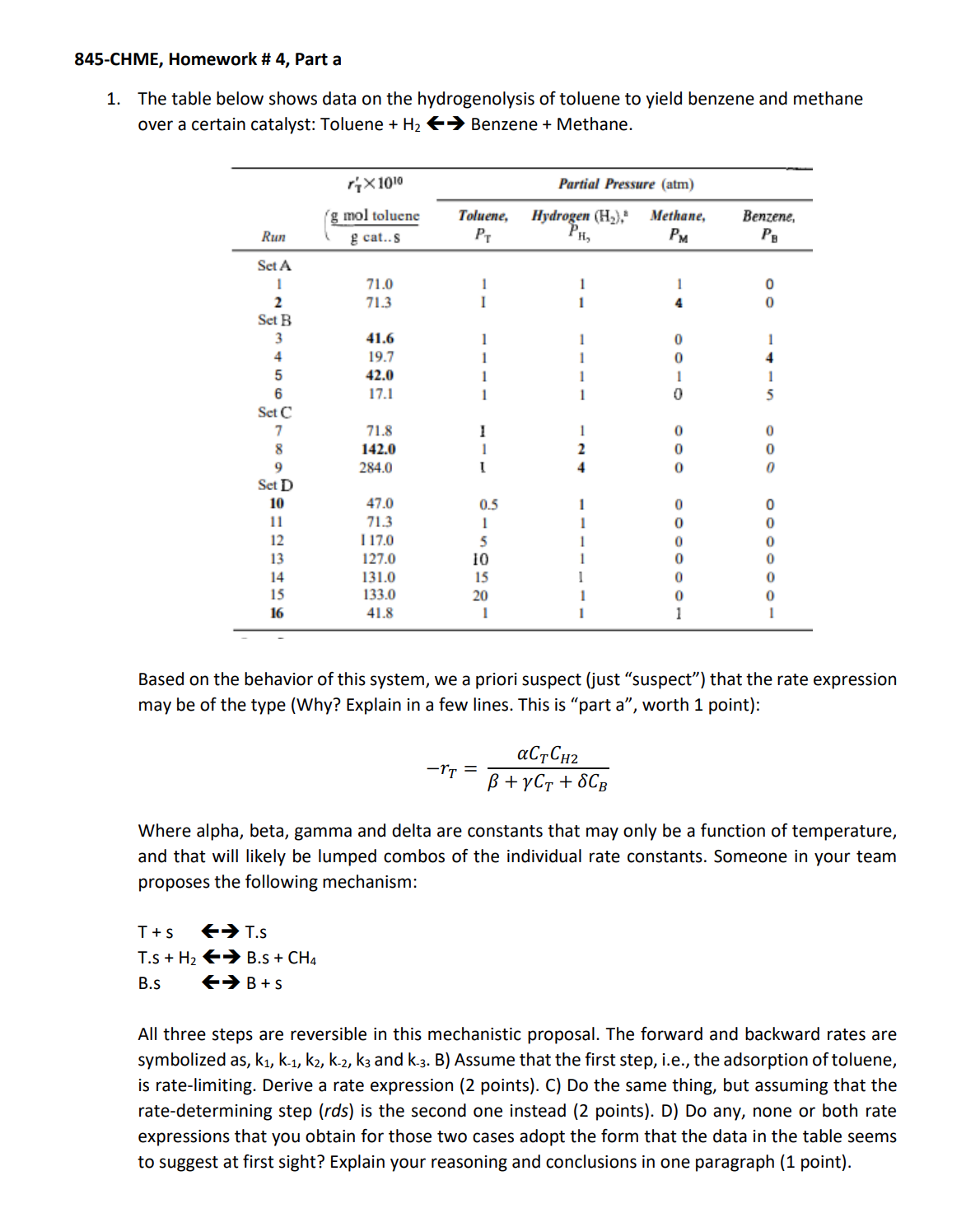
1. The table below shows data on the hydrogenolysis of toluene to yield benzene and methane over a certain catalyst: Toluene +H2 Benzene + Methane. Based on the behavior of this system, we a priori suspect (just "suspect") that the rate expression may be of the type (Why? Explain in a few lines. This is "part a", worth 1 point): rT=+CT+CBCTCH2 Where alpha, beta, gamma and delta are constants that may only be a function of temperature, and that will likely be lumped combos of the individual rate constants. Someone in your team proposes the following mechanism: T+sT.sT.s+H2B.s+CH4B.sB+s All three steps are reversible in this mechanistic proposal. The forward and backward rates are symbolized as, k1,k1,k2,k2,k3 and k3. B) Assume that the first step, i.e., the adsorption of toluene, is rate-limiting. Derive a rate expression (2 points). C) Do the same thing, but assuming that the rate-determining step (rds ) is the second one instead (2 points). D) Do any, none or both rate expressions that you obtain for those two cases adopt the form that the data in the table seems to suggest at first sight? Explain your reasoning and conclusions in one paragraph (1 point)
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


