Answered step by step
Verified Expert Solution
Question
1 Approved Answer
organic chemistry, fill chart 2. Which of the following molecules would you expect to dissolve in water? Diethyl ether? Briefly explain based on your understanding
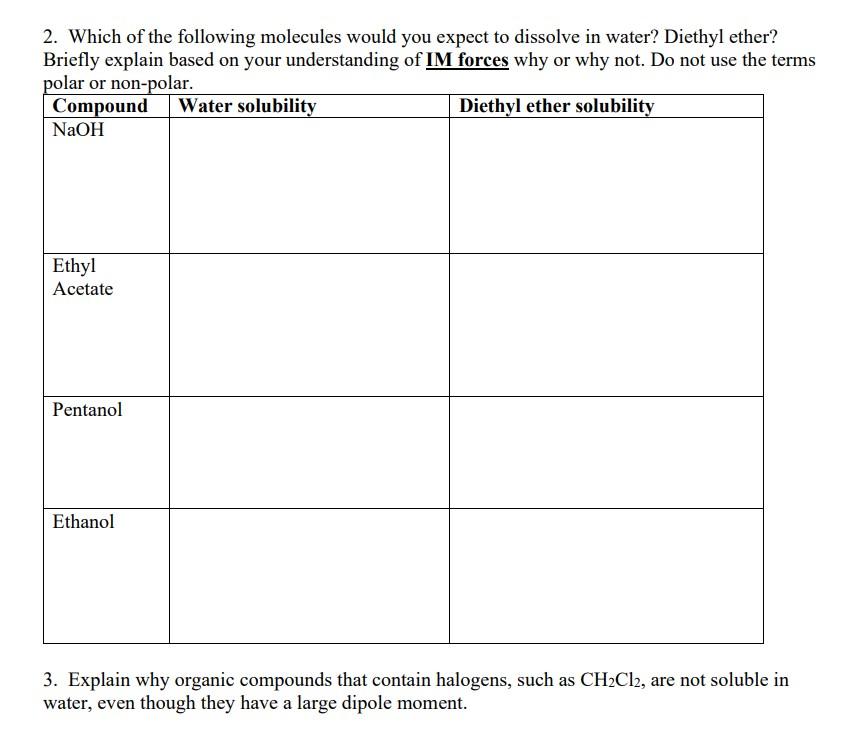
organic chemistry, fill chart
2. Which of the following molecules would you expect to dissolve in water? Diethyl ether? Briefly explain based on your understanding of IM forces why or why not. Do not use the terms 3. Explain why organic compounds that contain halogens, such as CH2Cl2, are not soluble in water, even though they have a large dipole momentStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


