Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please answer it all and show full explanation Consider the cell. Cu Cut (0.00803 M) || Ag+ (0.00573 M)|Ag = 0.521 V = 0.799 V
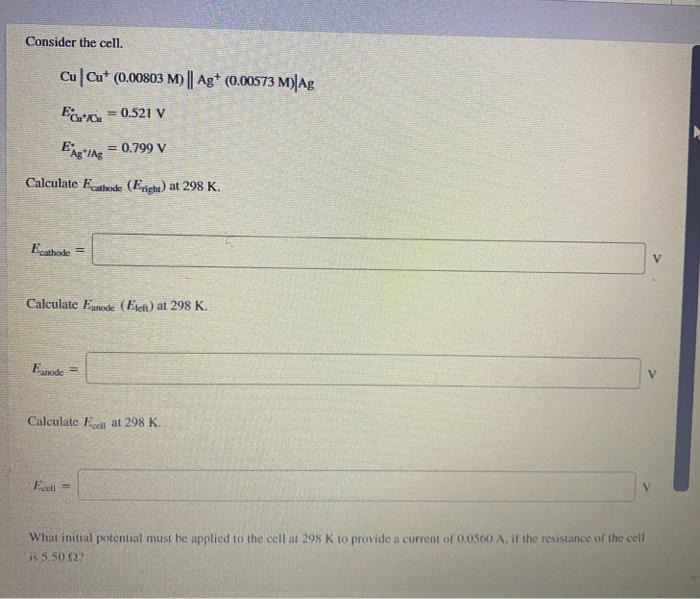
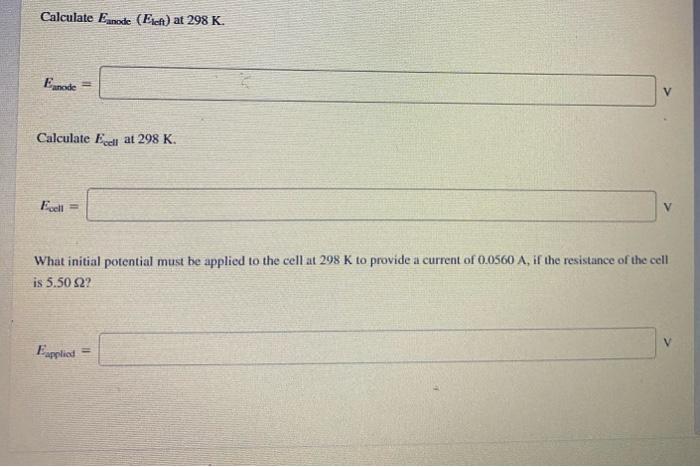
please answer it all and show full explanation
Consider the cell. Cu Cut (0.00803 M) || Ag+ (0.00573 M)|Ag = 0.521 V = 0.799 V Calculate cathode (Eright) at 298 K. Ecathode = Calculate Ennode (Eteft) at 298 K. Eanode Calculate Eclat 298 K. Fee What initial potential must be applied to the cell at 298 K to provide a current of 0.0500 At the resistance of the cell is 5.50522 Calculate Eanode (EA) at 298 K. Eanode = Calculate Eceli at 298 K. Ecell = What initial potential must be applied to the cell at 298 K to provide a current of 0.0560 A, if the resistance of the cell is 5.50 82? Eapplied Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


