Answered step by step
Verified Expert Solution
Question
1 Approved Answer
please explain in details Tc a = For a gas that obeys the van der Waals equation, the Te and Pe are given by the
please explain in details 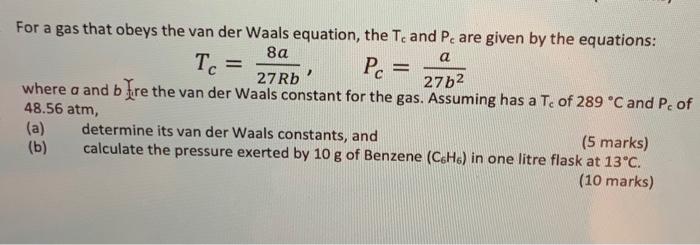
Tc a = For a gas that obeys the van der Waals equation, the Te and Pe are given by the equations: 8 Pc = 27 Rb 27b2 where a and b Fre the van der Waals constant for the gas. Assuming has a Te of 289 C and Pc of 48.56 atm, (a) determine its van der Waals constants, and (5 marks) (b) calculate the pressure exerted by 10 g of Benzene (C6H6) in one litre flask at 13C. (10 marks) 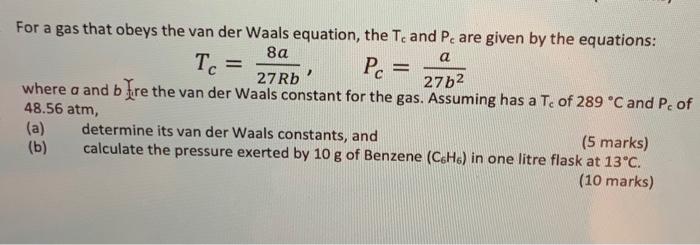
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


