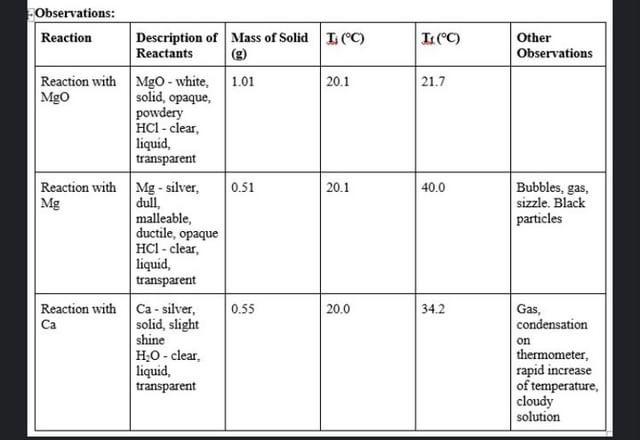
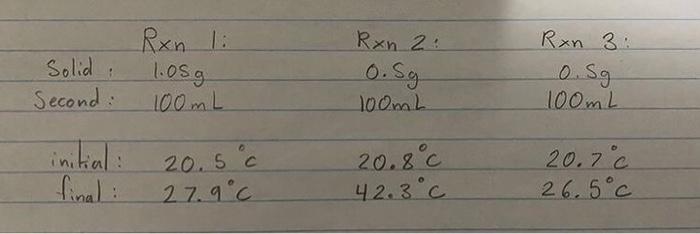Please help me with this lab (all questions are in order)
Experimental Applications of Hess's Law Purpose: The purpose of this lab is to use Hess's law to determine the enthalpy of reaction for reactions whose energy cannot be easily measured directly. Background: There are many reactions for which the enthalpy of reaction cannot be easily measured experimentally. Some of these are dangerous to perform in the lab. Others generate so much enthalpy that simple calorimeters cannot be used. In such cases, it is practical to examine a series of reactions whose net effect is the desired reaction, but whose enthalpys of reaction are more easily measured. Once that has been done, the required enthalpy of reaction can be calculated using Hess's Law. In the first investigation, the combustion of magnesium is examined. The burning of magnesium can be represented by the equation: Mg(s) + 10 Ma. Mgos Mg) + % 020) The above equation can be obtained by manipulating the three reactions shown below: 1. Mgo + 2 HCI) MgCl2 + H2O 2. Mga MgCl20) + 3. + 02/0) H2O (s) + 2 HC2) H219) H20 In the second investigation, the enthalpy of formation of solid calcium hydroxide is examined. The equation for the reaction is: Ca + 0.219) H219) Ca(OH)2 The above equation can be obtained by combining the following two reactions: 4. Ca + 2 H2O Ca(OH)2 + H2O 5. H2lo) + 02/01 H2O Magnesium and acid reactions, and the calcium and water reaction are measured experimentally. The Standard Enthalpies of Formation value is used for the formation of water and you will look up this value. **This lab uses the assumption that a dilute aqueous solution has the same density and specific heat capacity as water. Materials: Materials per group: Equipment styrofoam cups, 250 ml, 2 cardboard lid for cups, 1 thermometer, alcohol, 1 graduated cylinder, 100 mL, 1 glass stirring rod, 1 100 ml 100 ml Chemicals distilled water hydrochloric acid, HCH (1.0 mol/L) magnesium oxide, Mgom magnesium metal, ribbon, Mg. calcium metal, Can 19 0.59 0.59 Safety Precautions: PaviAMALLoafat Anand Amanding the Annapplicabla Procedure: The Enthalpy of Combustion of Magnesium: 1. Read through the procedure and create a neat data table to record your results. 2. Clear the desk and put on PPE. 3. Nest the 2 styrofoam cups together to create a rudimentary calorimeter. Use the cardboard square as a lid to insulate the calorimeter. Use one hole for the thermometer and one hole to allow reaction gases to escape. 4. Place 100 mL of 1.0 mol/L hydrochloric acid into the nested styrofoam cups calorimeter. Record the temperature of the solution 5. Accurately find (to the nearest 0.01 g) and record the mass of about 1 g of solid magnesium oxide. 6. Add the solid magnesium oxide to the acid solution in the cup. Stir the mixture gently with the thermometer TECORE Somorrature reached 9+ ... with the thermometer and record the highest temperature reached. 7. Discard the solution as instructed by the teacher and rinse the cup thoroughly with water. 8. Place 100 mL of 1.0 mol/L hydrochloric acid solution into the styrofoam cup. Record the temperature of the solution. 9. Accurately find (to the nearest 0.01 g) and record the mass of about 0.5 g of magnesium ribbon. 10. Add the magnesium ribbon to the acid solution. Stir the mixture gently with the thermometer and record the highest temperature reached. 11. Discard the solution as instructed by the teacher and rinse the cup thoroughly with water. The Enthalpy of Formation of solid Calcium Hydroxide: The Enthalpy of Formation of solid Calcium Hydroxide: 1. Clear the desk and put on PPE. 2. Place 100 mL of water into the styrofoam cup. Record the temperature of the water. 3. Accurately find (to the nearest 0.01 g) and record the mass of about 0.5 g of calcium metal. CAUTION: Do not allow any of the pieces of calcium metal to become wet. 4. Add the calcium to the water. 5. Stir the mixture and record the highest temperature reached. 6. Dispose of waste and clean up, as instructed by the teacher. Observations: Prepare a data table based on the procedure. Read it! You should include qualitative observations of reactants and the reaction(sounds), products, Observations: Prepare a data table based on the procedure. Read it! You should include qualitative observations of reactants and the reaction(sounds), products. Record required measurements. Temperatures should be recorded to one decimal place (estimated). Masses should be recorded to two decimal places. Units must be included. Analysis: The Enthalpy of Combustion of Magnesium: 1. Two reactions were carried out for this section. (Equations 1. and 2. from the background.) From the experimental data, calculate the following for EACH reaction. a. The temperature change of the solution in each case. b. The heat absorbed by the solution in each case. C. The number of moles magnesium oxide and magnesium used. d. The enthalpy change in kJ/mol for magnesium oxide reacting with hydrochlorica Analysis: The Enthalpy of Combustion of Magnesium: 1. Two reactions were carried out for this section. (Equations 1. and 2 from the background.) From the experimental data, calculate the following for EACH reaction. a. The temperature change of the solution in each case. b. The heat absorbed by the solution in each case. C. The number of moles magnesium oxide and magnesium used. d. The enthalpy change in kJ/mol for magnesium oxide reacting with hydrochloric acid. e. The enthalpy change in kJ/mol for magnesium reacting with hydrochloric acid. Show ALL calculations in a clear, well-communicated format. 2. Write out equations 1, 2 and 3 from the background' and beside each write the enthalpy change for that reaction. Enthalpy change values for equations 1. and 2. were calculated in question #1. above and you will need to look up the value for equation 3 3. Apply Hess's Law. Using equations 1, 2 and 3 (written above) as your data equations', manipulate them to determine the enthalpy change for the combustion of magnesium, the 'destination equation'. Your answer' is the molar enthalpy for the combustion of magnesium (also enthalpy of formation of magnesium). Include a final statement. 4. Look up the standard enthalpy of combustion of magnesium (also enthalpy of formation of magnesium) from a reliable source and state the value and the source. Calculate the percentage error between the accepted and experimental(your calculated) values for the enthalpy of combustion of magnesium. The Enthalpy of Formation of Solid Calcium Hydroxide: 1. One reaction was carried out for this section. From the data for the reaction calculate the following: a The temperaturament The Enthalpy of Formation of Solid Calcium Hydroxide: 1. One reaction was carried out for this section. From the data for the reaction, calculate the following: a. The temperature change of the solution. b. The heat absorbed by the solution. c. The number of moles calcium used. d. The enthalpy of reaction in kJ/mol for calcium reacted with water. Show ALL calculations in a clear, well-communicated format. 2. Write out equations 4 and 5 from the background' and beside each write the enthalpy change for that reaction. The enthalpy change value for equation 4. was calculated in question #1. above and you will need to look up the value for equation 5. 3. Apply Hess's Law. Using equations 4 and 5 (written above) as your data equations', manipulate them to determine the enthalpy change for the formation of calcium hydroxide, the 'destination equation'. Your answer is the molar enthalpy of formation for calcium hydroxide. Include a final statement 4. Look up the standard enthalpy of formation of calcium hydroxide from a reliable source and state the value and the source. Calculate the percentage error between the accepted and experimental(your calculated) values for the molar enthalpy of formation of calcium hydroxide. Summary and Conclusion: 1. Referring to your percentage error calculations, would you say the lab was successful? Explain your answer. 2. Describe at least THREE sources of error in this lab. These should be detailed (2-3 sentences) and relate to your final result. (Does the error increase or decrease your final result or is it random?) They should NOT include 'mistakes" like spilling a reactant or copying down a value incorrectly. 3. Describe at least TWO possible ways to improve the final results (accuracy) of this lab. These should be things that we could do in our lab. (2-3 sentences each) Solid Second Ron li 1.05g 100mL Rxn 2 O. Sg 100mL Rxn 3: 0. Sg 100mL 20.5c final: 20.8c 42.3C 20.7C 26.5C 27.9C I(C) Other Observations 21.7 0.51 40.0 Observations: Reaction Description of Mass of Solid 1 (C) Reactants Reaction with MgO - white, 1.01 20.1 Mgo solid, opaque, powdery HCI-clear liquid, transparent Reaction with Mg - silver, 20.1 Mg dull malleable, ductile, opaque HCI-clear liquid, transparent Reaction with Ca - silver, 0.55 20.0 Ca solid, slight shine H:0 - clear liquid, transparent Bubbles, gas, sizzle. Black particles 34.2 Gas, condensation on thermometer, rapid increase of temperature, cloudy solution