Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please show solution! Thanks! Enter your answer in the provided box. Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30108at700C:2H2S(g)2H2(g)+S2(g) If 0.39 mol
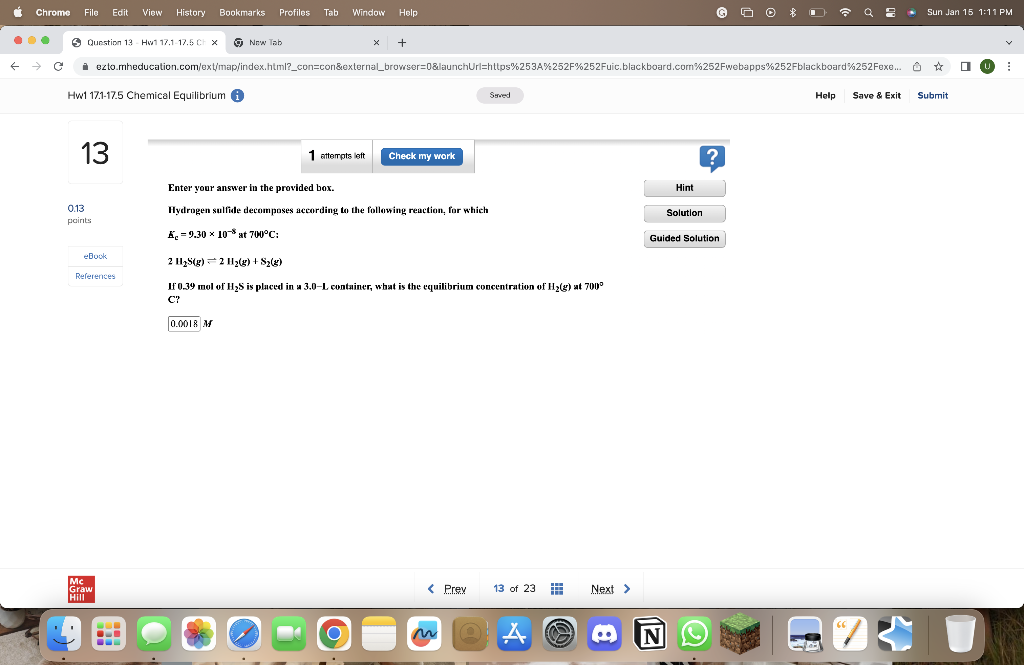
Please show solution! Thanks!
Enter your answer in the provided box. Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30108at700C:2H2S(g)2H2(g)+S2(g) If 0.39 mol of H2S is pluced in a 3.0L container, what is the equilibrium concentration of H2(g) at 700 C? MStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


