Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Please solve this problem for a student to understand not for expert. Sometimes the answer is wrong. Please be careful. I will give you like
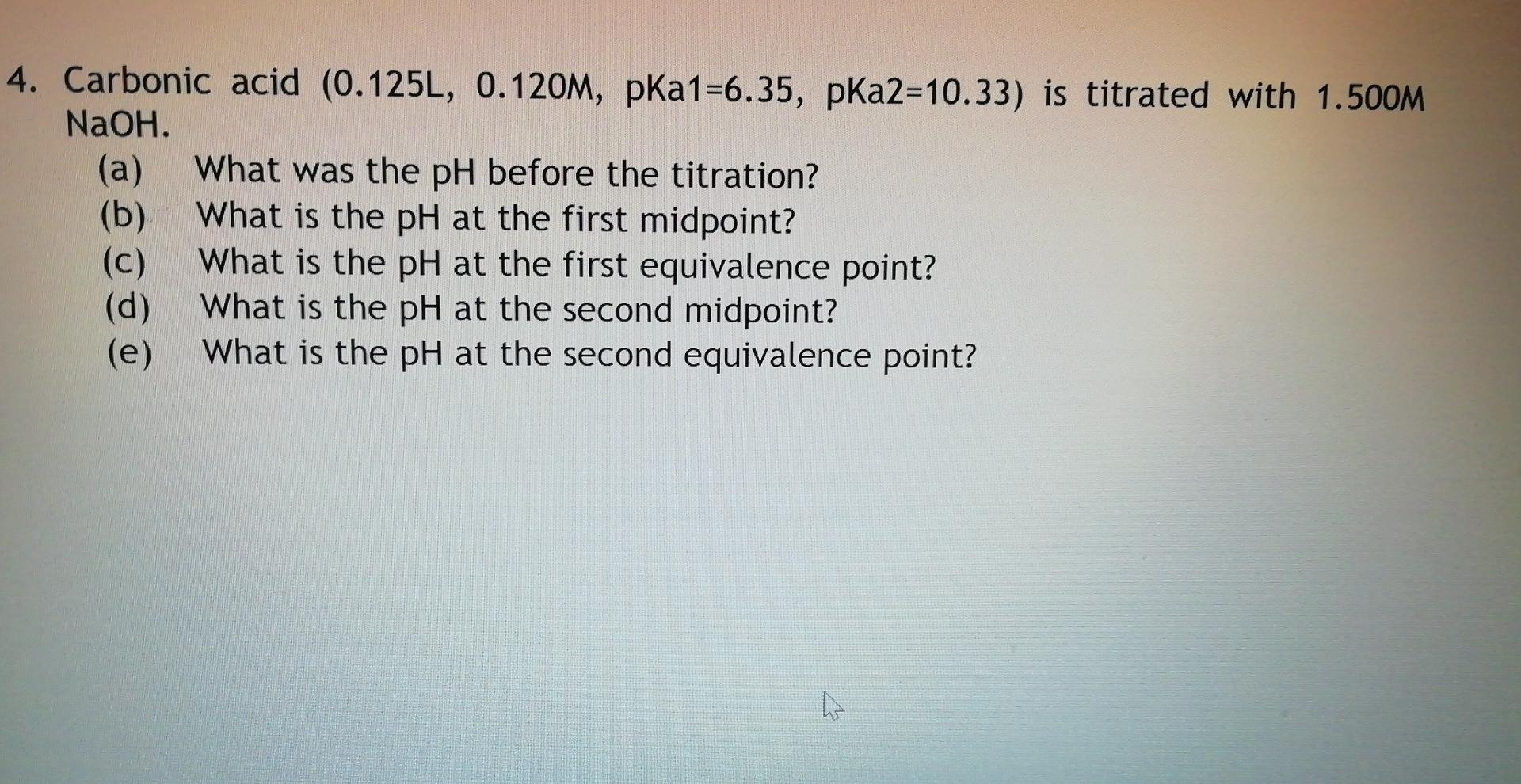
Please solve this problem for a student to understand not for expert. Sometimes the answer is wrong. Please be careful. I will give you like for you solution. Thanks!
4. Carbonic acid (0.125L, 0.120M, pka1=6.35, pka2=10.33) is titrated with 1.500M NaOH. (a) What was the pH before the titration? (b) What is the pH at the first midpoint? (c) What is the pH at the first equivalence point? (d) What is the pH at the second midpoint? (e) What is the pH at the second equivalence point? WStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


