Answered step by step
Verified Expert Solution
Question
1 Approved Answer
plz answer all, wil give What is the molar mass of your molecular formula, C2H4O2? (Enter your answer to two decimal places.) 62.037 g/mol Submit
plz answer all, wil give 
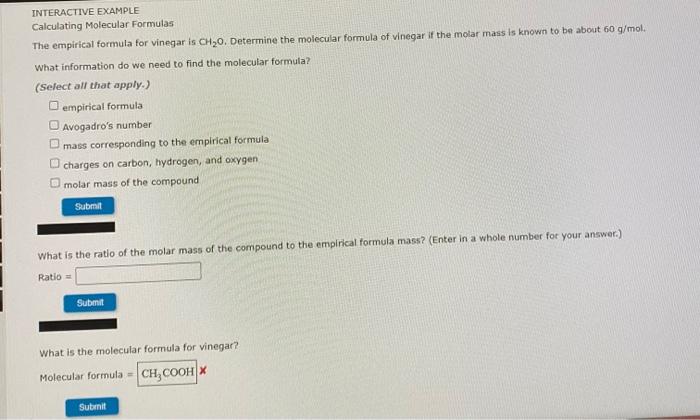
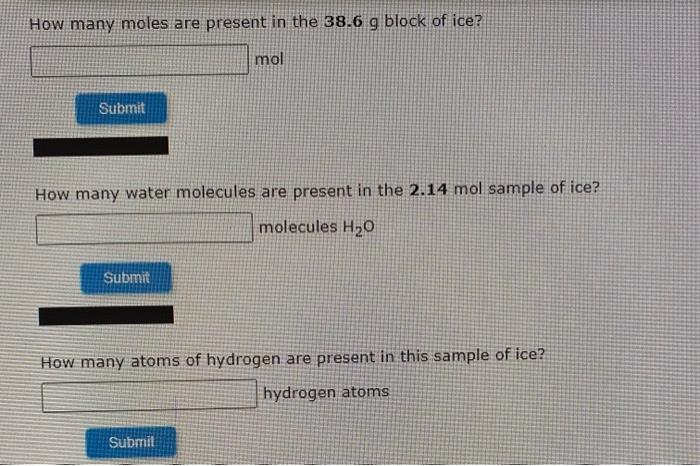
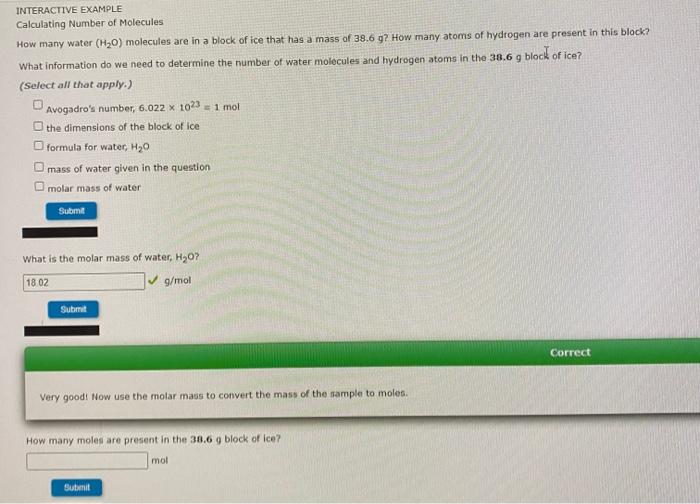
What is the molar mass of your molecular formula, C2H4O2? (Enter your answer to two decimal places.) 62.037 g/mol Submit INTERACTIVE EXAMPLE Calculating Molecular Formulas The empirical formula for vinegar is CH20. Determine the molecular formula of vinegar if the molar mass is known to be about 60 g/mol. What information do we need to find the molecular formula? (Select all that apply.) empirical formula Avogadro's number mass corresponding to the empirical formula charges on carbon, hydrogen, and oxygen molar mass of the compound Submit What is the ratio of the molar mass of the compound to the empirical formula mass? (Enter in a whole number for your answer.) Ratio = Submit What is the molecular formula for vinegar? Molecular formula = CH COOH Submit How many moles are present in the 38.6 g block of ice? mol Submit How many water molecules are present in the 2.14 mol sample of ice? molecules H20 Submit How many atoms of hydrogen are present in this sample of ice? hydrogen atoms Submit INTERACTIVE EXAMPLE Calculating Number of Molecules How many water (H20) molecules are in a block of ice that has a mass of 38.6 g? How many atoms of hydrogen are present in this block? What information do we need to determine the number of water molecules and hydrogen atoms in the 30.6 g block of ice? (Select all that apply.) Avogadro's number, 6.022 x 1023 = 1 mol the dimensions of the block of foe formula for water, H20 mass of water given in the question molar mass of water Submi What is the molar mass of water, H307 18.02 g/mol Submit Correct Very good. Now use the molar mass to convert the mass of the sample to moles. How many moles are present in the 38,6 g block of ice? mol Sutimit 



Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


