Answered step by step
Verified Expert Solution
Question
1 Approved Answer
plz can you solve it? 2 Known gas sample has a volume of 710 mmHg and 450 mL at 27c. what would the volume (mL)
plz can you solve it? 
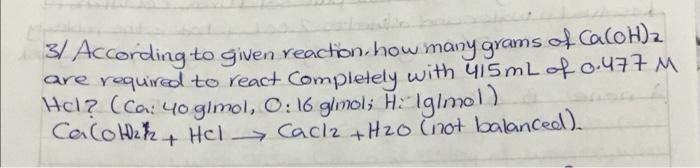

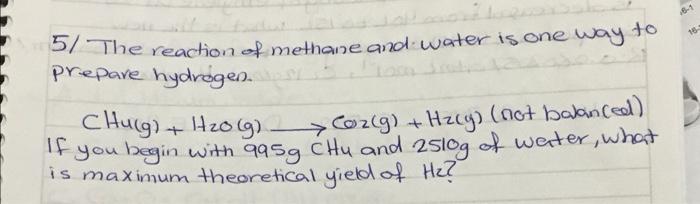
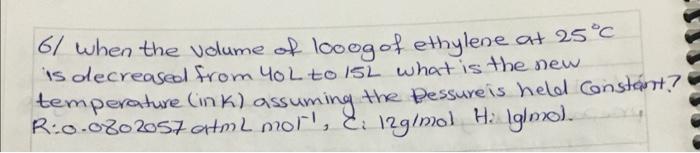

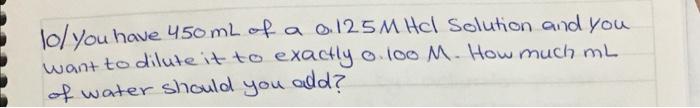
2 Known gas sample has a volume of 710 mmHg and 450 mL at 27c. what would the volume (mL) of the gas be if it measured at Standard temperature and pressure (STP)? CR:0-6802057 athlma tki'; R: 0-08314s bar 2 mei's') 3/ According to given reaction how many grams of CaCOH)2 2 are required to react Completely with 415mL of 0.477 M Hel? (Ca: 40 gimol, O: 16 glinals Hulgimol) Calotah Hel Cacl2 + H2O (not balanced) . + 4/ when loo g of the 90% pure solid urea, COWHz)2, is dissolved in water and olikuted to 2.00L. what is the motorty of the solution? (0:12 gmol, Hilgimol, Ox16 g/mol) Nugimol. 5/ The reaction of methane and water is one way to prepare hydrogen. CHulg) + H20lg) Corg) + Hzcy) (not balanced) -you begin with 995g CHu and 2510g of water, what is maximum theoretical yield of He?? + () ) IC 6/ when the volume of loog of ethylene at 25C is decreased from you to 15L what is the new temperature (ink) assuming the bessure is held constant? ? R:0.0802057 atm L moll, E. 12g/mol Ha Iglmo). al Density of aluminum is 2.7 g/cm3 Calculate the mass of a aluminum block has lo cm X 8mm x 9cm dimension (AI: 26.989/mol) lol you have 450 mL of a 0125M Hcl Solution and you want to dilute it to exactly o. 100 M. How much ml of water should you add 
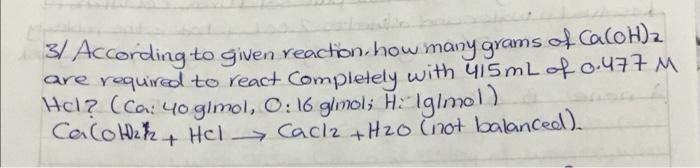

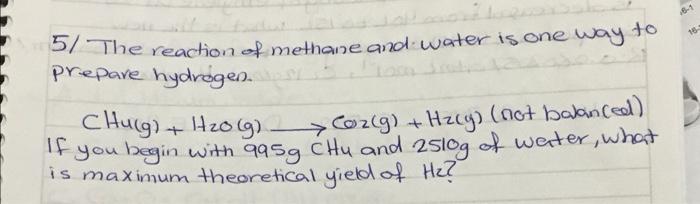
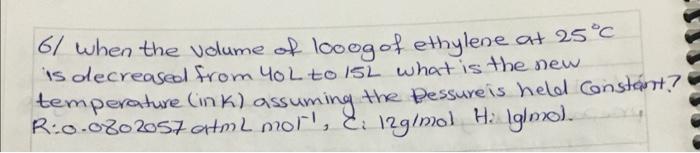

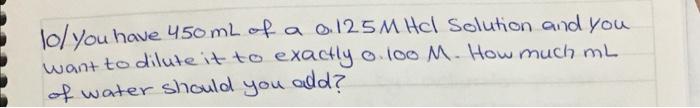
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


