Question
Problem #1: This compound only contains carbons and hydrogens. A. Determine the M peak (Which m/z peak represents the M peak). B. Determine the

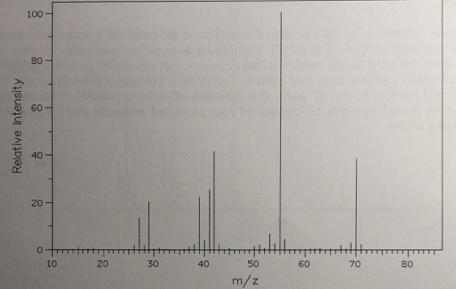
Problem #1: This compound only contains carbons and hydrogens. A. Determine the M peak (Which m/z peak represents the M peak). B. Determine the chemical formula. Determine the degrees of unsaturation. What functional group does this compound contain? C. Identify the structure for the M" peak and the base peak (tallest peak). Hint: Look for the most stable carbocation. 100 80 60 40 10 30 40 50 60 70 80 m/z Relative Intensity 20
Step by Step Solution
3.40 Rating (150 Votes )
There are 3 Steps involved in it
Step: 1
Answer A The M Peak is at mz 70 B The chemical formula is C 5 H 10 Because the M peak su...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Organic Chemistry
Authors: Marc Loudon
5th edition
981519431, 978-0981519449, 098151944X, 978-0-98151943, 978-0981519432
Students also viewed these Chemistry questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



