Problem 3: Felix and Company reports the following information. (1) Use the high-low method to estimate...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
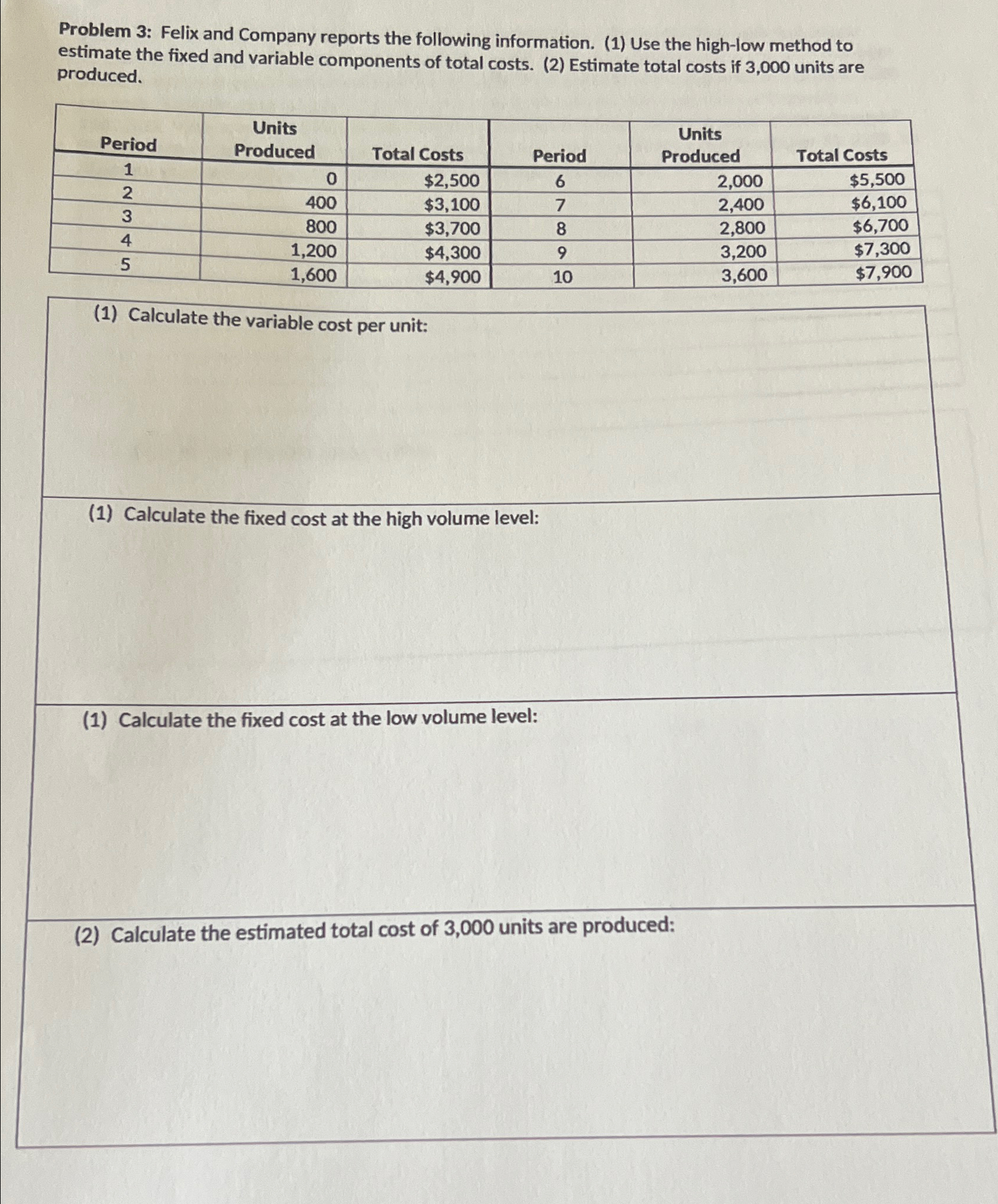
Problem 3: Felix and Company reports the following information. (1) Use the high-low method to estimate the fixed and variable components of total costs. (2) Estimate total costs if 3,000 units are produced. Period Units Produced Units Total Costs Period Produced Total Costs 1 0 $2,500 6 2,000 $5,500 2 400 $3,100 7 2,400 $6,100 3 800 $3,700 8 2,800 $6,700 4 1,200 $4,300 9 3,200 $7,300 5 1,600 $4,900 10 3,600 $7,900 (1) Calculate the variable cost per unit: (1) Calculate the fixed cost at the high volume level: (1) Calculate the fixed cost at the low volume level: (2) Calculate the estimated total cost of 3,000 units are produced: Problem 3: Felix and Company reports the following information. (1) Use the high-low method to estimate the fixed and variable components of total costs. (2) Estimate total costs if 3,000 units are produced. Period Units Produced Units Total Costs Period Produced Total Costs 1 0 $2,500 6 2,000 $5,500 2 400 $3,100 7 2,400 $6,100 3 800 $3,700 8 2,800 $6,700 4 1,200 $4,300 9 3,200 $7,300 5 1,600 $4,900 10 3,600 $7,900 (1) Calculate the variable cost per unit: (1) Calculate the fixed cost at the high volume level: (1) Calculate the fixed cost at the low volume level: (2) Calculate the estimated total cost of 3,000 units are produced:
Expert Answer:

Posted Date:
Students also viewed these accounting questions
-
A companys retained earnings on December 31, 2018 was $2,190,000 and its shareholders equity was $8,760,000. During 2019 the company reported the following: Net income $225,000 A sale of treasury...
-
A language school owner takes an SRS of 10 of the 72 Introductory Spanish classes offered by the school. Each student in each of the sampled classes is given a vocabulary test and is also asked...
-
Multiply. (2a+1)(7a+4)
-
A zoologist recorded the weight, in grams, for a sample of white-fronted Amazon parrots. The following MINITAB output presents the results of a hypothesis test regarding the mean weight of this...
-
In divisional income statements prepared for Brooks Construction Company, the Payroll Department costs are charged back to user divisions on the basis of the number of payroll checks, and the...
-
Matheson Electronics has just developed a new electronic device that it believes will have broad market appeal. The company has performed marketing and cost studies that revealed the following...
-
Customers buy cars from three auto companies. Given the company from which a customer last bought a car, the probability that she will buy her next car from each company is as follows: a If someone...
-
What does John Hale's testimony tell you about the social environment insalem?
-
How does Apple company's iPod is playing a strategic role for the company?
-
Skyler Manufacturing recorded operating data for its shoe division for the year. Sales Contribution margin Controllable fixed costs Average total operating assets $4,500,000 500,000 200,000 900,000...
-
To help finance a major expansion, Large Hadron Company sold a noncallable bond 5 years ago that now has 10 years to maturity. This bond has a 8.25% annual coupon, paid semiannually, sells at a price...
-
Assuming that the following tax rate schedule applies, what is your tax liability given ordinary taxable income of $87,400? 10% ($0-$9,875) .12% ($9,875-$40,125) 22% ($40,125-$85,525) .24%...
-
Multiple Choice Questions: 1) What is the ?h value for the reverse reaction of the following reaction: co2 (g) + 2h2o (l) ? ch4 (g) + o2 (g) ?hforward = +890 kj a. + 445 kj b. 445 kj c. + 890 kj d. ...
-
A sample statistic will not change from sample to sample. Determine whether the statement is true or false. If it is false, rewrite it as a true statement.
-
Applying the criterion for equilibrium, derive the Clausius-Clapeyron equation.
-
A binary liquid mixture consists of \(60 \mathrm{~mol}\) per cent ethylene and \(40 \mathrm{~mol}\) per cent propylene. At \(423 \mathrm{~K}\), the vapour pressure of ethylene and propylene are...
-
For the system methanol (1)-methyl acetate (2), the activity coefficients for components 1 and 2 are represented by where \[ \begin{aligned} \ln \gamma_{1} & =A x_{2}^{2} \quad \text { and } \quad...

Study smarter with the SolutionInn App

