Answered step by step
Verified Expert Solution
Question
1 Approved Answer
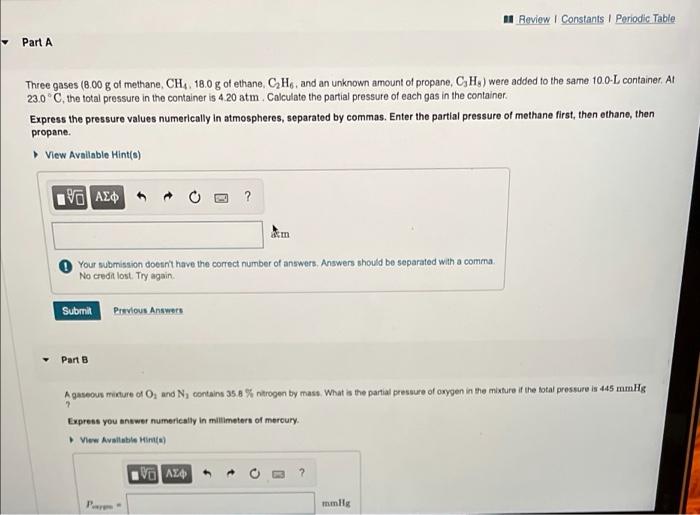
Pt. 1 Three gases (8.00 g g of methane, C H 4 C H 4 , 18.0 g g of ethane, C 2 H 6
Pt. 1 Three gases (8.00
g
g
of methane,
C
H
4
C
H
4
, 18.0
g
g
of ethane,
C
2
H
6
C
2
H
6
, and an unknown amount of propane,
C
3
H
8
C
3
H
8
) were added to the same 10.0-
L
L
container. At 23.0
C
C
, the total pressure in the container is 4.20
atm
a
t
m
. Calculate the partial pressure of each gas in the container.
Express the pressure values numerically in atmospheres, separated by commas. Enter the partial pressure of methane first, then ethane, then propane.
pt. 2
A gaseous mixture of
O
2
O
2
and
N
2
N
2
contains 35.8
%
%
nitrogen by mass. What is the partial pressure of oxygen in the mixture if the total pressure is 445
mmHg
m
m
H
g
?
Express you answer numerically in millimeters of mercury.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


