Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Q 1 ) Show that work done during the vaporization of 1 k g of saturated liquid water at 2 0 0 C can be
Q Show that work done during the vaporization of of saturated liquid water at can be calculated in two different approaches, and calculate the work done using both approaches.
Q A well insulated gas cylinder contains air initially at bars and Calculate the maximum work that can be obtained by opening the cylinder valve. Assume air to be an ideal gas with a constant specific heat capacity of Surroundings temperature and pressure are and bar, respectively.
Q Nitrogen gas is being withdrawn at a rate of from a cylinder. Initially, the pressure of the gas in the cylinder is bar and its temperature is The cylinder is wellinsulated. What will be the temperature and pressure of the gas in the cylinder after minutes? Assume Nitrogen is an ideal gas with a constant specific heat capacity.
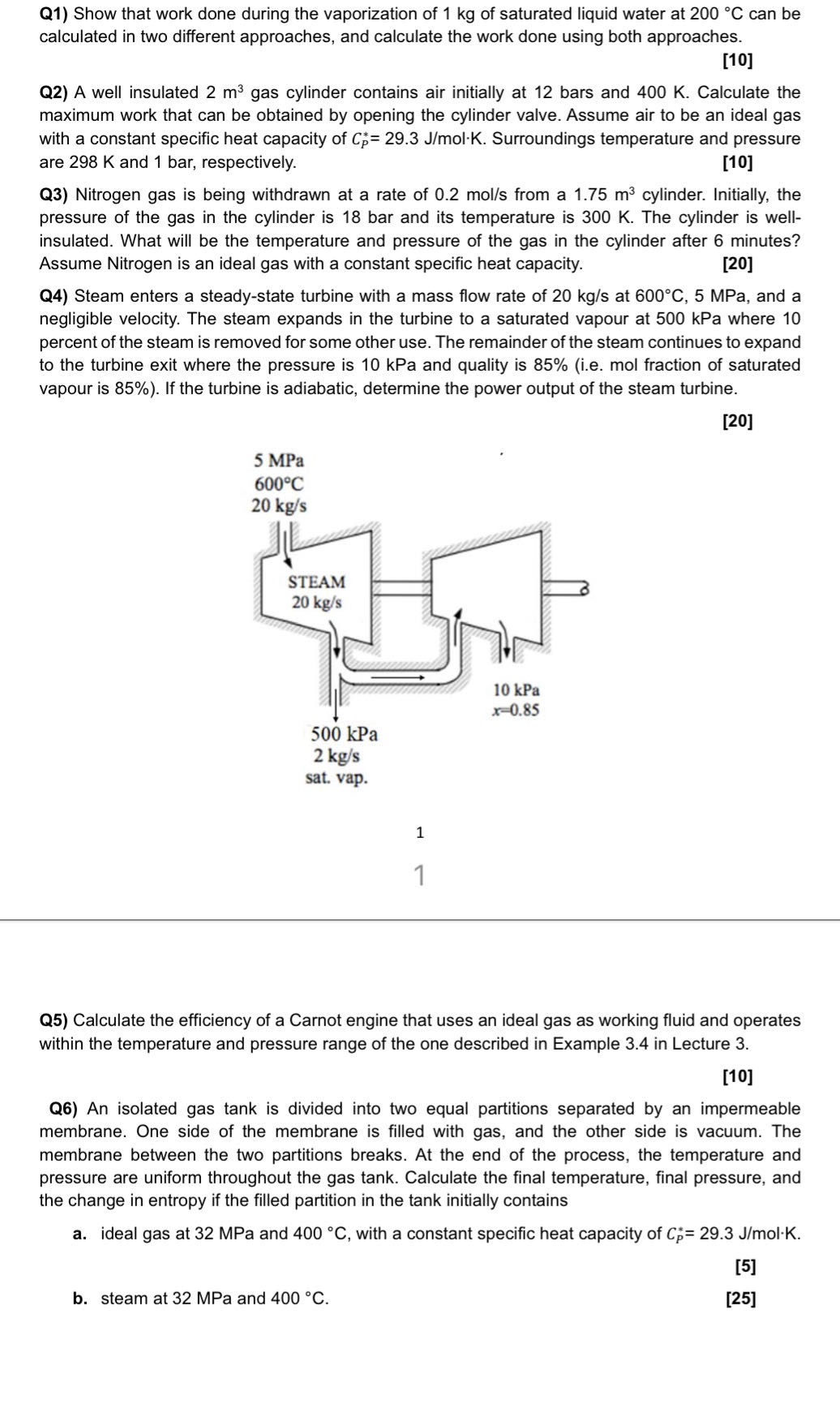
Q Steam enters a steadystate turbine with a mass flow rate of at MPa, and a negligible velocity. The steam expands in the turbine to a saturated vapour at kPa where percent of the steam is removed for some other use. The remainder of the steam continues to expand to the turbine exit where the pressure is kPa and quality is ie mol fraction of saturated vapour is If the turbine is adiabatic, determine the power output of the steam turbine.
Q Calculate the efficiency of a Carnot engine that uses an ideal gas as working fluid and operates within the temperature and pressure range of the one described in Example in Lecture
Q An isolated gas tank is divided into two equal partitions separated by an impermeable membrane. One side of the membrane is filled with gas, and the other side is vacuum. The membrane between the two partitions breaks. At the end of the process, the temperature and pressure are uniform throughout the gas tank. Calculate the final temperature, final pressure, and the change in entropy if the filled partition in the tank initially contains
a ideal gas at MPa and with a constant specific heat capacity of
b steam at MPa and
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


