Answered step by step
Verified Expert Solution
Question
1 Approved Answer
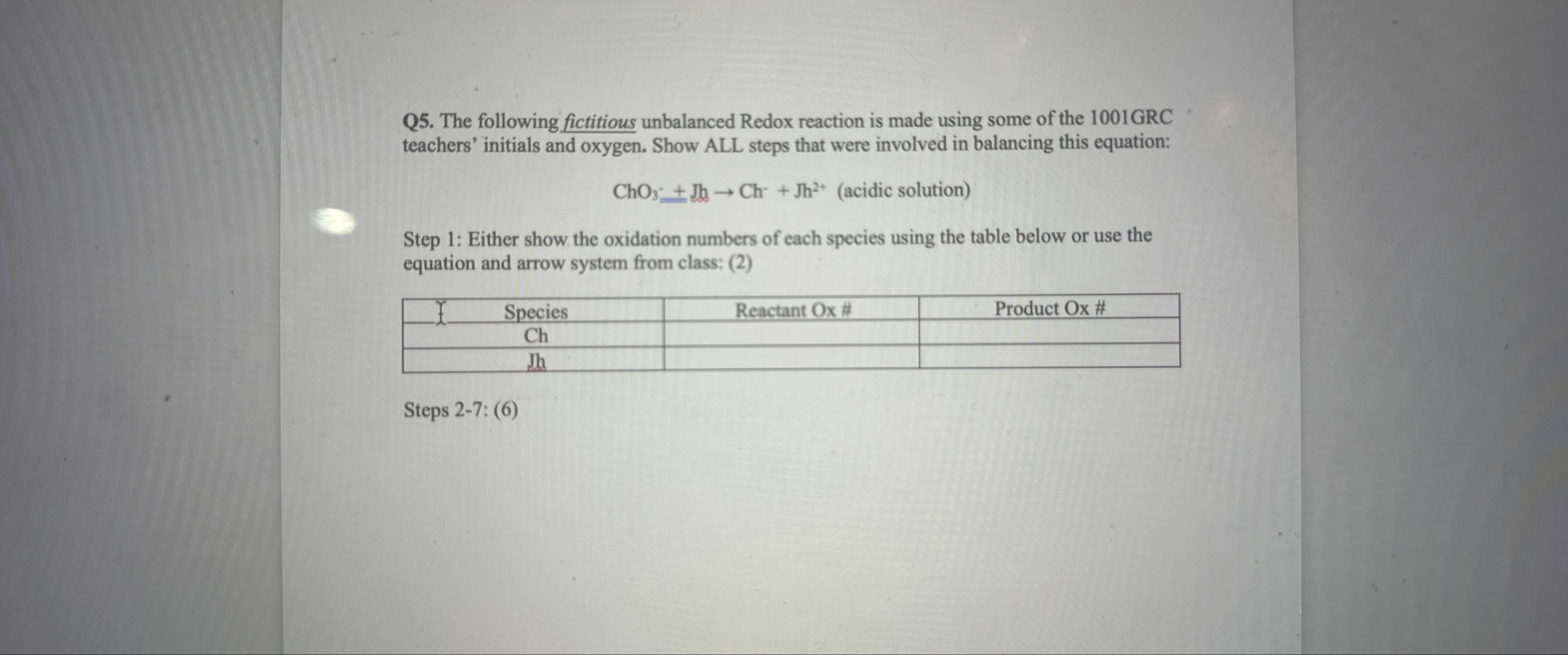
Q 5 . The following fictitious unbalanced Redox reaction is made using some of the 1 0 0 1 GRC teachers' initials and oxygen. Show
Q The following fictitious unbalanced Redox reaction is made using some of the GRC teachers' initials and oxygen. Show ALL steps that were involved in balancing this equation:
solution
Step : Either show the oxidation numbers of each species using the table below or use the equation and arrow system from class:
tableSpeciesReactant #Product Ox#
Steps :
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


