Answered step by step
Verified Expert Solution
Question
1 Approved Answer
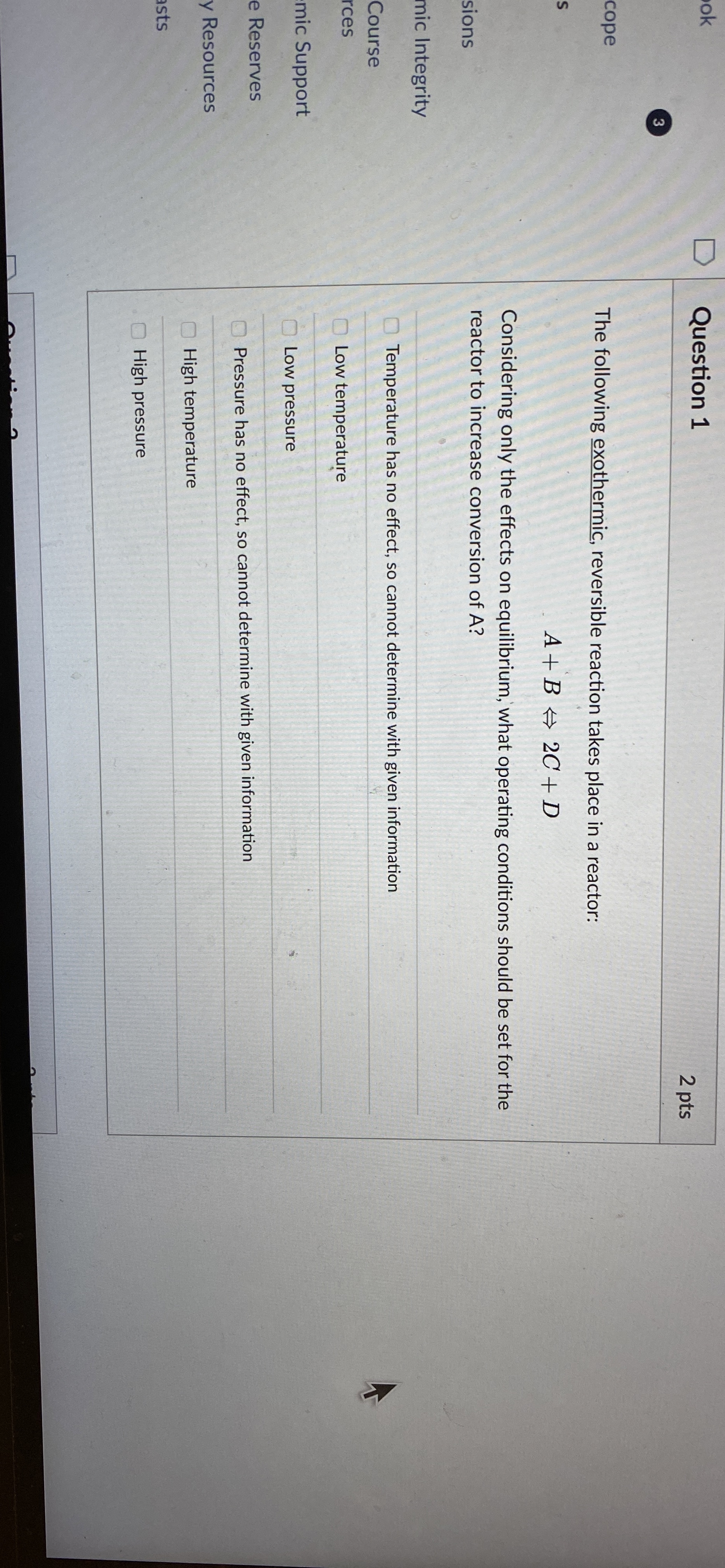
Question 1 3 cope The following exothermic, reversible reaction takes place in a reactor: S sions mic Integrity Course res mic Support e Reserves y
Question
cope
The following exothermic, reversible reaction takes place in a reactor:
S
sions
mic Integrity
Course
res
mic Support
e Reserves
y Resources
sts
Considering only the effects on equilibrium, what operating conditions should be set for the reactor to increase conversion of
Temperature has no effect, so cannot determine with given information
Low temperature
Low pressure
Pressure has no effect, so cannot determine with given information
High temperature
High pressure
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


