Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Question 9 4 pts When the methyl ester of 4-oxopentanoic acid (methyl 4-oxopentanoate) is reacted with MeMgBr the product would contain which of the
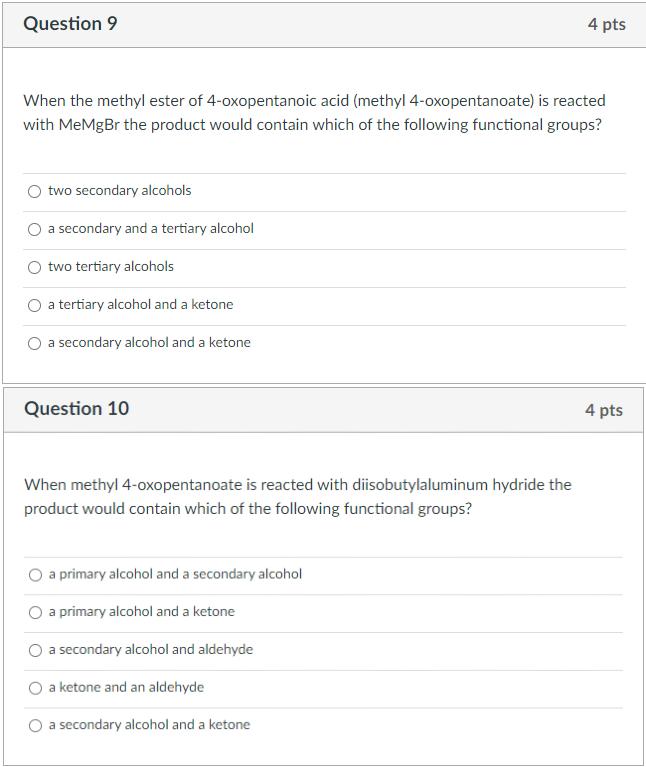
Question 9 4 pts When the methyl ester of 4-oxopentanoic acid (methyl 4-oxopentanoate) is reacted with MeMgBr the product would contain which of the following functional groups? two secondary alcohols a secondary and a tertiary alcohol two tertiary alcohols a tertiary alcohol and a ketone a secondary alcohol and a ketone Question 10 4 pts When methyl 4-oxopentanoate is reacted with diisobutylaluminum hydride the product would contain which of the following functional groups? a primary alcohol and a secondary alcohol a primary alcohol and a ketone a secondary alcohol and aldehyde a ketone and an aldehyde a secondary alcohol and a ketone
Step by Step Solution
★★★★★
3.46 Rating (153 Votes )
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Document Format ( 2 attachments)
6363d0b282e2a_238726.pdf
180 KBs PDF File
6363d0b282e2a_238726.docx
120 KBs Word File
Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


