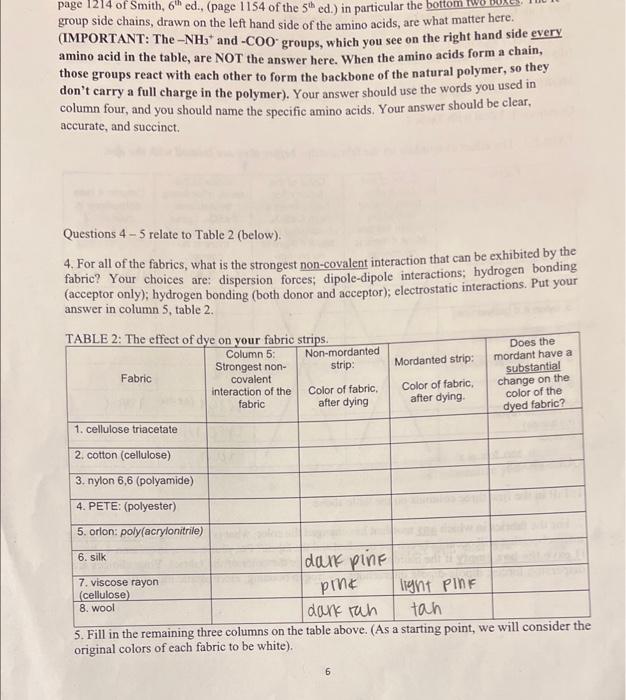
Questions 1-3 deal with Table 1. TABLE 1: The effect of mordant on the fabric strips before they are treated with the dye. QUESTIONS: 1: Fill in the first three columns on the table above. 2. For the fabrics in which any mordant has changed the color, what is the strongest non-covalent interaction that will occur between the mordant and the fabric? Put your answer in column 4 above. (Leave the box empty if the mordant does not change the color of the fabric). Your choices are: dispersion forces; dipole-dipole interactions; hydrogen bonding; electrostatic interactions. group side chains, drawn on the left hand side of the amino acids, are what matter here. (IMPORTANT: The NH3+and COOgroups, which you see on the right hand side every amino acid in the table, are NOT the answer here. When the amino acids form a chain, those groups react with each other to form the backbone of the natural polymer, so they don't carry a full charge in the polymer). Your answer should use the words you used in column four, and you should name the specific amino acids. Your answer should be clear. accurate, and succinct. Questions 45 relate to Table 2 (below). 4. For all of the fabrics, what is the strongest non-covalent interaction that can be exhibited by the fabric? Your choices are: dispersion forces; dipole-dipole interactions; hydrogen bonding (acceptor only); hydrogen bonding (both donor and acceptor); electrostatic interactions. Put your answer in column 5 , table 2 . 5. Fill in the remaining three columns on the table above. (As a starting point, we win consiuet une original colors of each fabric to be white). Questions 1-3 deal with Table 1. TABLE 1: The effect of mordant on the fabric strips before they are treated with the dye. QUESTIONS: 1: Fill in the first three columns on the table above. 2. For the fabrics in which any mordant has changed the color, what is the strongest non-covalent interaction that will occur between the mordant and the fabric? Put your answer in column 4 above. (Leave the box empty if the mordant does not change the color of the fabric). Your choices are: dispersion forces; dipole-dipole interactions; hydrogen bonding; electrostatic interactions. group side chains, drawn on the left hand side of the amino acids, are what matter here. (IMPORTANT: The NH3+and COOgroups, which you see on the right hand side every amino acid in the table, are NOT the answer here. When the amino acids form a chain, those groups react with each other to form the backbone of the natural polymer, so they don't carry a full charge in the polymer). Your answer should use the words you used in column four, and you should name the specific amino acids. Your answer should be clear. accurate, and succinct. Questions 45 relate to Table 2 (below). 4. For all of the fabrics, what is the strongest non-covalent interaction that can be exhibited by the fabric? Your choices are: dispersion forces; dipole-dipole interactions; hydrogen bonding (acceptor only); hydrogen bonding (both donor and acceptor); electrostatic interactions. Put your answer in column 5 , table 2 . 5. Fill in the remaining three columns on the table above. (As a starting point, we win consiuet une original colors of each fabric to be white)