Question
Regarding the following configuration, which of the following statements is true and explain why: A. It has two (2) gauche interactions. B. It does NOT
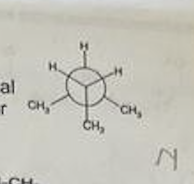
Regarding the following configuration, which of the following statements is true and explain why: A. It has two (2) gauche interactions. B. It does NOT exhibit steric strain. C. It exhibits torsional strain. D. It exhibits angular strain. 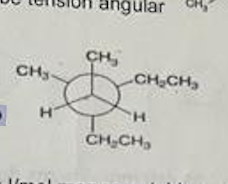
The IUPAC name of the following molecule island explain why: A. diethyl dimethylethane B. 2,2-diethylbutane C. 3-sec-butylpentane D. 3-ethyl-3-methylpentane.
If the eclipsed conformation of CH3CH2CI is 15 kJ/mol less stable than its staggered conformation, then the value of an eclipsed H-CI interaction is: A. 3.8 kJ/mol B. 7 kJ/mol C. 7.6 kJ/mol D. 15 kJ/mol. and why?
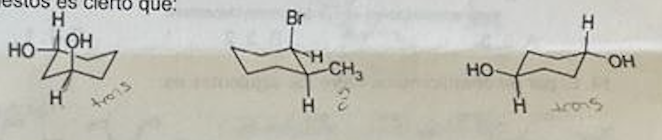
Regarding the following compounds, it is true that and why? A.all have trans geometry B.all have cis geometry C.only the one on the left is cis D. only the one in the middle is NOT trans Among the following isomers, the one that has the lowest energy conformer is and why?: A. cis-1,2-dimethylcyclohexane B. trans-1,2-dimethylcyclohexane C. cis-1,3-dimethylcyclohexane D. cis-1,4-dimethylcyclohexane
estus ciemo queStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


