Answered step by step
Verified Expert Solution
Question
1 Approved Answer
show the movement of electrons by half arrows for the following reaction to the OH oxygen atom of cumyl hydroperoxide causing the adjacent OO petoxide
show the movement of electrons by half arrows for the following reaction 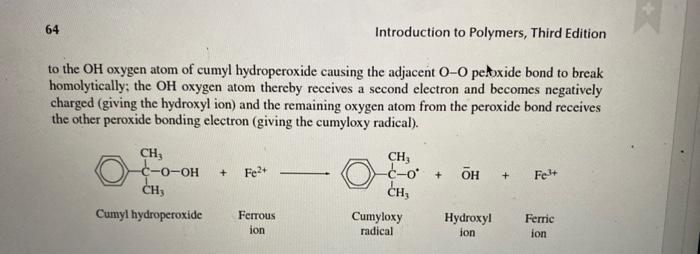
to the OH oxygen atom of cumyl hydroperoxide causing the adjacent OO petoxide bond to break homolytically; the OH oxygen atom thereby receives a second electron and becomes negatively charged (giving the hydroxyl ion) and the remaining oxygen atom from the peroxide bond receives the other peroxide bonding electron (giving the cumyloxy radical) 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


