Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Study the model below. Answer the following questions. Question 13 (1 point) The electron pair geometry of this molecule is linear trigonal planar tetrahedral The
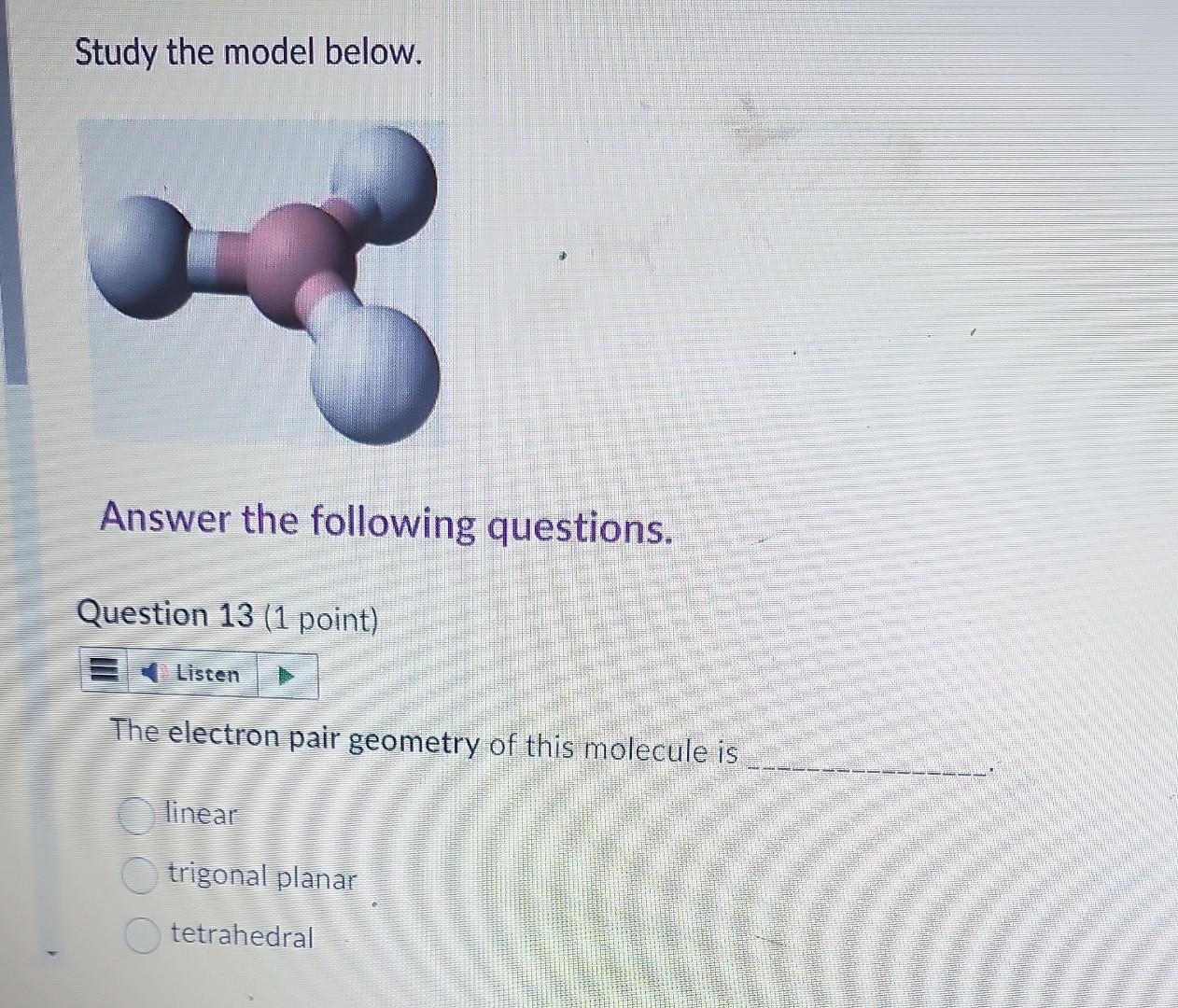
Study the model below. Answer the following questions. Question 13 (1 point) The electron pair geometry of this molecule is linear trigonal planar tetrahedral The molecular shape of this molecule is linear trigonal planar tetrahedral trigonal pyramidal V-shaped Question 15 (1 point) How many lone electron pair(s) is/are around the central atom? 0 1 2 The bond angle is 104.5107109.5120
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


