Temperature C 200 150 100 50 0 -13.9 HO Vapor + Syrup 20 Syrup (liquid) 62.4 Ice + Sugar 40 60 wt% Sugar 185
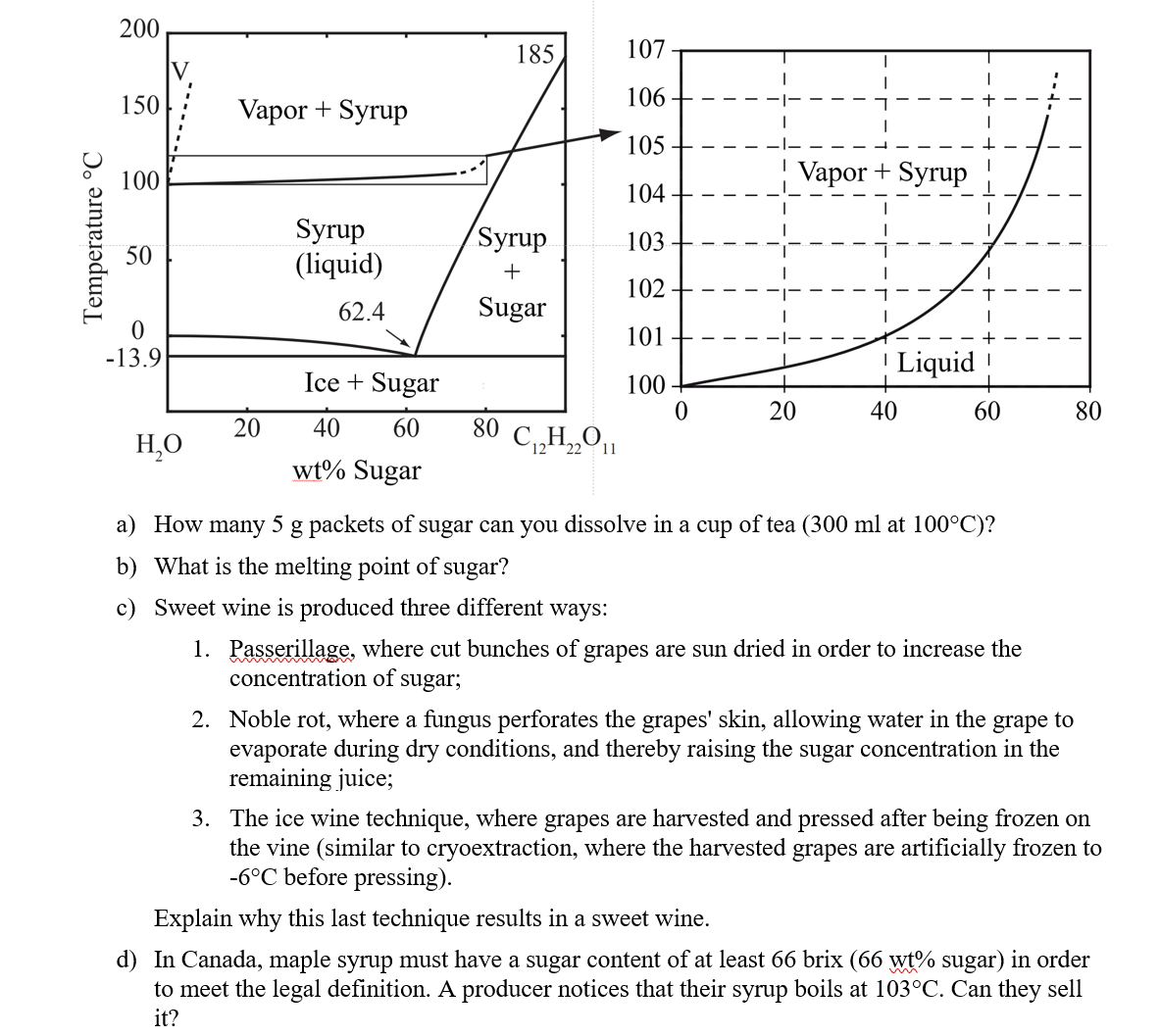
Temperature C 200 150 100 50 0 -13.9 HO Vapor + Syrup 20 Syrup (liquid) 62.4 Ice + Sugar 40 60 wt% Sugar 185 Syrup + Sugar 80 CH2O11 22 11 107 106 105 104 103 102 101 100 0 20 Vapor + Syrup ! Liquid ! 40 60 a) How many 5 g packets of sugar can you dissolve in a cup of tea (300 ml at 100C)? b) What is the melting point of sugar? c) Sweet wine is produced three different ways: 1. Passerillage, where cut bunches of grapes are sun dried in order to increase the concentration of sugar; I I 1 80 2. Noble rot, where a fungus perforates the grapes' skin, allowing water in the grape to evaporate during dry conditions, and thereby raising the sugar concentration in the remaining juice; 3. The ice wine technique, where grapes are harvested and pressed after being frozen on the vine (similar to cryoextraction, where the harvested grapes are artificially frozen to -6C before pressing). Explain why this last technique results in a sweet wine. d) In Canada, maple syrup must have a sugar content of at least 66 brix (66 wt% sugar) in order to meet the legal definition. A producer notices that their syrup boils at 103C. Can they sell it?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get step-by-step solutions from verified subject matter experts
100% Satisfaction Guaranteed-or Get a Refund!
Step: 2Unlock detailed examples and clear explanations to master concepts

Step: 3Unlock to practice, ask and learn with real-world examples

See step-by-step solutions with expert insights and AI powered tools for academic success
-
 Access 30 Million+ textbook solutions.
Access 30 Million+ textbook solutions.
-
 Ask unlimited questions from AI Tutors.
Ask unlimited questions from AI Tutors.
-
 Order free textbooks.
Order free textbooks.
-
 100% Satisfaction Guaranteed-or Get a Refund!
100% Satisfaction Guaranteed-or Get a Refund!
Claim Your Hoodie Now!

Study Smart with AI Flashcards
Access a vast library of flashcards, create your own, and experience a game-changing transformation in how you learn and retain knowledge
Explore Flashcards





