Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The drawings below illustrate four mixtures of two diatomic gases (nitrogen, N 2 (blue) and oxygen, O 2 (red)) in flasks of identical volume and
The drawings below illustrate four mixtures of two diatomic gases (nitrogen, N2 (blue) and oxygen, O2 (red)) in flasks of identical volume and at the same temperature.
(a) Which flask has the highest partial pressure of oxygen, depicted by the red molecules?
(b) Which flask has the lowest partial pressure of nitrogen, depicted by the blue molecules?
(c) Which flask does the Partial Pressure of O2 equal the Partial Pressure of N2?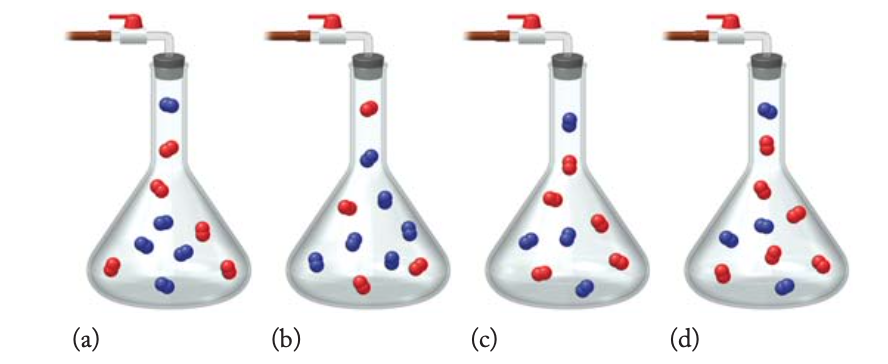
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


