Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The figure below shows a hot gas stream at 500 C being cooled to 300 C by transferring heat to the liquid water that
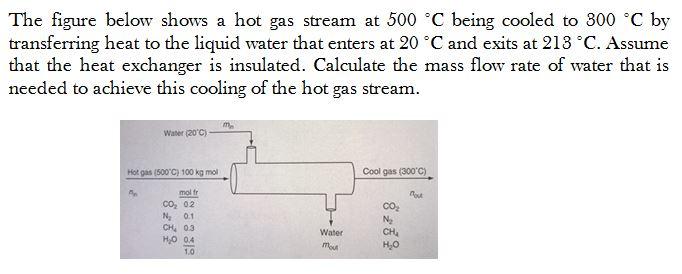
The figure below shows a hot gas stream at 500 C being cooled to 300 C by transferring heat to the liquid water that enters at 20 C and exits at 218 C. Assume that the heat exchanger is insulated. Calculate the mass flow rate of water that is needed to achieve this cooling of the hot gas stream. Water (20'C) Hot gas (500 C) 100 kg mol Cool gas (300'C) mol fr out co, 02 N 01 CH 03 Water CH, vo o'H 1.0
Step by Step Solution
★★★★★
3.41 Rating (154 Votes )
There are 3 Steps involved in it
Step: 1
Solution If the hot gas inlet str...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


